The Athlete Heart Mystery: Why Hard Work Looks Like “Rust” but Keeps You Safe
-
The Big Surprise: When Healthy Hearts Look Sick
For decades, doctors and runners shared a comfortable belief: if you ran marathons, your heart was bulletproof. This was known as the “Bassler hypothesis,” the idea that finishing a 26-mile race gave you a “get out of jail free” card against heart disease. However, as modern heart scans improved, doctors were met with a shock. The very people who were the fittest on the planet—lifelong marathoners and cyclists—often showed more “rust” or buildup in their heart pipes (arteries) than people who sat on the couch.
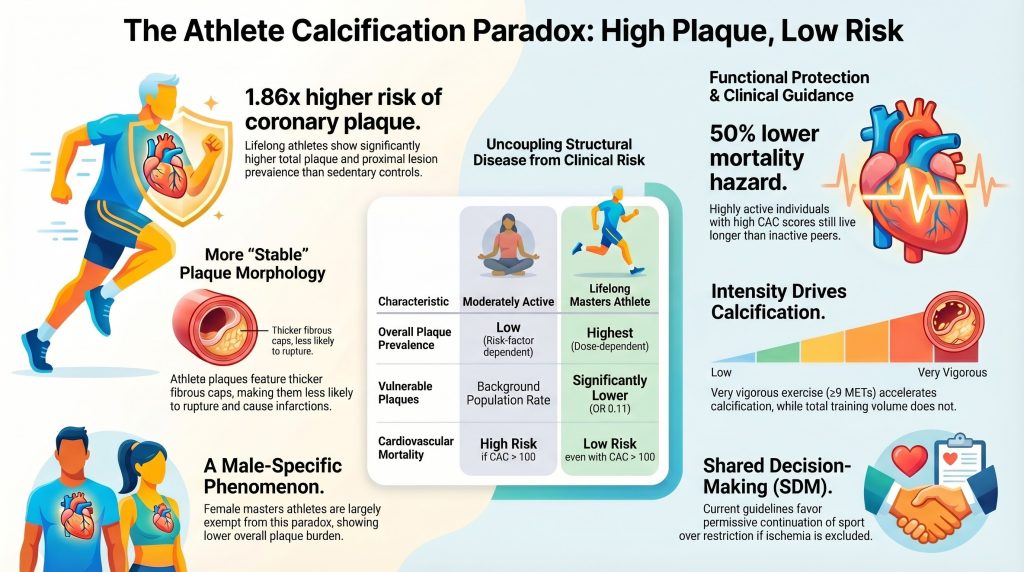
This discovery is called the Athlete Calcification Paradox. Scientists originally thought exercise acted as a perfect shield, keeping heart pipes smooth and clear. Instead, scans proved that many veteran athletes have high levels of calcium, a marker we usually associate with aging and heart attacks. This changed how we think: we now know that athletes aren’t immune to artery buildup; rather, their bodies handle that buildup in a way that is totally different from everyone else.
So What? This change in thinking means doctors can’t just look at a scan and tell a runner to “stop.” We now have to look at whether that buildup is a sign of a disease or a sign of a heart that has simply worked very hard.
Connective Tissue: To solve this mystery, we have to look at the actual data from thousands of athletes to see why “rusty” pipes aren’t always a sign of a failing engine.
-
The Evidence: What the Big Studies Found
To understand a mystery this big, scientists looked at large groups of people to find the truth. Three major studies showed us that an athlete’s heart pipes don’t look like a normal person’s.
- The Heinz Nixdorf Recall Study: Researchers looked at 108 male marathon runners. Even though these men were very fit, they had higher “calcium scores” (buildup) than regular people who had the same low risk for heart problems. It proved that running doesn’t just “wash away” gunk.
- The UK Masters Study: This study found that while male athletes had more buildup than people who sat on the couch, the type was different. Athletes had “hard” buildup, while the couch-sitters had more “soft” buildup, which is much more dangerous.
- The Master@Heart Study: This study looked at lifelong athletes and found they had more plaque (heart pipe rust) than even people who started exercising later in life. However, it also found that these athletes were 89% less likely to have the kind of dangerous, “pop-able” plaques that cause heart attacks.
Comparing the Heart Pipes: Athletes vs. Inactive People
| Category | Sedentary People | Lifelong Athletes |
| Overall Buildup (Plaque) | Lower overall | The more you exercise, the more you have |
| High-Risk “Pop-able” Spots | More common | 89% less likely |
| Overall Death Risk | Higher | Lowest |
So What? This data tells us that having “rust” in the pipes doesn’t mean the “engine” is going to fail. In an athlete, the buildup isn’t a sign of a clog—it’s often a sign that the pipe has been reinforced.
Connective Tissue: While we know athletes have more buildup, the way you exercise actually changes how much “rust” your body decides to create.
-
Hard vs. Long: How Exercise Intensity Changes Your Heart Pipes
Not all exercise is the same when it comes to the heart. For a long time, we thought it was just the number of hours you spent training (volume) that caused these changes. However, the MARC-2 study found a different “trigger.”
It turns out that “very vigorous” exercise—training so hard that you can’t hold a conversation (9 METs or higher)—is what actually speeds up the calcium buildup. Interestingly, “regular vigorous” exercise (6 to 9 METs), where you are working hard but can still talk, is actually a “sweet spot” that seems to protect the heart and leads to less buildup.
When you push your heart to its absolute limit, blood rushes through your pipes with incredible force. This creates “mechanical stress,” acting like a tiny hammer hitting the inside of your pipes. To protect itself from this hammering, the body “patches” the area with calcium to make the walls stronger. This is a biological repair job, not a disease.
So What? This means the “rust” is actually a protective shield created by your body to handle the stress of going “all-out.”
Connective Tissue: This “patching” process is the secret reason why an athlete’s heart stays safer than a regular person’s heart.
-
Why Athlete “Rust” is Different: The Stable Plaque Secret
In the heart, “hard” buildup is much safer than “soft” buildup. Doctors worry about “soft” buildup because it is like a thin-skinned blister; if it pops, it causes a heart attack. “Hard” buildup is like a scar—it is tough, stable, and very unlikely to break.
Athletes grow a biological shield (which scientists call “neo-media”) over their buildup. This is a new, tough inner wall that locks the gunk away so it can’t cause trouble.
Beyond this shield, an athlete’s heart makes three clever safety changes:
- Wider Pipes: The heart pipes actually grow, leaving extra room inside the tube. A 50% blockage in a massive pipe still leaves plenty of room for blood to flow.
- Backup Pipes: Athletes grow “collateral circulation”—a web of tiny extra pipes that can take over if a main pipe ever gets blocked.
- Turbo-Charging Ability: Athletes have a better “pumping reserve,” meaning their pipes can open wider and move extra blood exactly when the heart needs it most.
So What? This is why a “50% blockage” in a runner is much less dangerous than in someone who doesn’t exercise. The runner has a bigger pipe, a turbo-charged pump, and a biological shield protecting the area.
Connective Tissue: The ultimate proof of this safety isn’t found in a scan, but in the survival rates of these athletes.
-
The Scoreboard: Who Lives Longer?
The most important data point isn’t what a scan looks like—it’s whether people actually live longer. The Cooper Center Study looked at over 21,000 men and found that even athletes with high calcium scores (over 100) were much less likely to die than inactive people with the same scores.
Interestingly, female athletes don’t seem to have this paradox at all. Their hearts stay much clearer of buildup than men’s. Scientists believe this is because of estrogen (a hormone that protects the heart) and the fact that women’s bodies have a less “angry” inflammatory response to the stress of hard exercise.
So What? The big takeaway is that the benefits of exercise “dominate” the risks of the buildup. You are much better off exercising and having some “rust” than sitting on the couch and having none.
Connective Tissue: Because exercise is so protective, the rules for how doctors treat athletes are being rewritten.
-
The New Rules for Doctors and Athletes
Doctors are moving away from telling athletes to “stop.” According to the 2025 AHA/ACC Rules, the goal is now to keep people moving safely. Here are the “New Rules”:
- Shared Decision-Making: Doctors and athletes should talk about the risks together, balancing the scan results with the athlete’s goals and lifestyle.
- Keep Playing: If the athlete passes a stress test and their heart is strong, the doctor should actively encourage them to keep exercising rather than just “allowing” it.
- Use Medicine: High fitness doesn’t mean you are “too healthy” for help. Statins (cholesterol medicine) and blood pressure pills are still vital tools to keep heart pipes stable.
- Be Ready: Every team or race should have a “heart-starter” machine (AED) nearby and people who know CPR, just in case.
So What? These rules ensure that athletes aren’t “scared off” the field unnecessarily. They allow athletes to keep doing what they love while using modern medicine to stay safe.
Connective Tissue: These new rules reflect our final understanding of how the athlete’s heart truly works.
-
Conclusion: The Final Word on the Athlete Paradox
The mystery of the athlete’s heart is finally solved. The “rust” we see on scans isn’t a sign of a failing heart; it is a sign of a heart that has adapted to win. The Athlete Calcification Paradox shows us that intense exercise triggers a form of biological repair, creating a stronger, more stable system.
While the buildup might look scary on a computer screen, it is actually a shield. Staying active remains the single best thing you can do for your life. Even if your heart pipes look a little “rusty,” that rust is a sign of a durable engine built to go the distance.
DEEP DIVE
The Exercise Paradox: Why Lifelong Endurance Athletes Have More Coronary Plaque — and Still Live Longer
What the latest research on masters athletes, coronary artery calcification, and cardiovascular mortality tells us — and what it means for your heart health.
Picture two men walking into a cardiology clinic on the same day. The first has barely exercised in a decade. The second has run more than 40 marathons and still logs 60 miles a week. Both are in their mid-fifties. Both have identical coronary artery calcium scores — a measure of calcified plaque in the heart arteries — that their cardiologist flags as concerning. Which man is at greater risk?
If you answered the first man, you’d be right. And that answer gets to the heart of one of the most surprising discoveries in modern cardiology: athletes who train intensely for decades develop more calcium deposits in their coronary arteries than sedentary people — but they live longer anyway.
This is not a typo or a misunderstanding. It is a genuine, well-documented, and deeply studied paradox that researchers now call the “athlete calcification paradox.” Understanding it matters enormously — not just for competitive athletes, but for anyone trying to make sense of the relationship between exercise, heart disease, and longevity.
| Key Takeaway: Lifelong endurance athletes accumulate more coronary plaque than sedentary people — including non-calcified, high-risk plaque types — yet their risk of dying from heart disease remains substantially lower. This page explains why, and what it means for your health decisions. |
A Little History: From Invincibility to Complexity
For most of the 20th century, the medical community assumed that serious athletes were essentially immune to coronary artery disease. In the 1970s, a physician named Thomas Bassler published what became known as the “Bassler hypothesis” — the idea that completing a standard marathon conferred near-absolute protection against fatal coronary atherosclerosis.1 His argument captured the cultural moment. The running boom was in full swing, and the idea that the sport’s devotees had somehow transcended normal cardiovascular aging was enormously appealing.
That optimism began to unravel quickly. By the late 1970s and early 1980s, autopsy studies began documenting coronary atherosclerosis in marathon runners who had died suddenly during training or racing.2 These were not sedentary men with decades of poor habits — they were committed athletes whose arterial walls told a more complicated story than anyone expected.
Today, the scientific picture has shifted considerably. We now know that athletes are absolutely not immune to coronary artery disease. In fact, the latest evidence suggests something almost counterintuitive: certain kinds of extreme, lifelong exercise may actually accelerate the formation of coronary plaque, even as the same exercise dramatically reduces the risk of dying from it.
Who Are Masters Athletes?
Throughout this article, you’ll see the term “masters athlete.” In sports medicine and cardiology research, this refers to men and women over the age of 35 to 40 who have engaged in prolonged, systematic endurance training for a large portion of their adult lives.3 Think of your neighbor who has been running 50 miles a week since their twenties, or the person at your gym who still competes in triathlons at 58. These are masters athletes.
What makes this population scientifically fascinating is the apparent contradiction between what we’d expect from their lifestyle — lean, low-risk, metabolically healthy — and what imaging studies consistently find in their coronary arteries. Understanding this contradiction has become one of the most productive areas of inquiry in contemporary sports cardiology.
The Evidence: What the Research Has Found
Several major research programs have spent the past two decades systematically imaging the hearts of aging endurance athletes. Their findings have changed the field — and they tell a nuanced story that deserves to be understood properly.
The Heinz Nixdorf Recall Marathon Study: The First Big Wake-Up Call
One of the earliest studies to formally quantify this paradox evaluated 108 healthy male marathon runners aged 50 and older — men who had each completed at least five full marathons in the prior three years.4 Researchers expected that their hearts would look pristine. What they found was more complicated.
The runners’ median Framingham Risk Score — a standard tool for estimating heart disease probability based on blood pressure, cholesterol, age, and smoking — was just 7%, compared to 11% in a group of age-matched men from the general population. By traditional measures, the runners looked like the lower-risk group by a significant margin.
But when researchers measured coronary artery calcium (CAC) — a score that reflects the amount of calcified plaque in the heart arteries, with higher scores indicating more disease — the picture shifted. The runners’ median CAC score was statistically similar to the general age-matched population, and significantly higher than controls who were matched to them on risk-factor level rather than just age. Twelve percent of the runners who underwent specialized cardiac MRI showed signs of myocardial late gadolinium enhancement — an indicator of previous microscopic cardiac injury — and all of the coronary events that occurred during the follow-up period happened exclusively in runners with CAC scores of 100 or above.4
| What This Means
Standard risk calculators like the Framingham Risk Score consistently underestimate the true plaque burden in highly trained athletes. A runner can have a “low-risk” score on paper while carrying a meaningfully elevated calcium burden in their arteries. This is one of the most important clinical lessons from this body of research. |
The MARC Studies: Understanding Volume, Intensity, and Progression
A Dutch research program called the MARC study (Measuring Athlete’s Risk of Cardiovascular Events) has contributed some of the most detailed data we have on how exercise characteristics relate to coronary atherosclerosis in aging athletes.
The first phase, MARC-1, cross-sectionally evaluated 284 male athletes aged 45 and older using both a calcium scoring scan and a more detailed coronary CT angiography.5 The results confirmed what the Heinz Nixdorf data suggested: athletes who accumulated the highest lifelong exercise volumes — more than 2,000 MET-minutes per week, a measure of energy expenditure — had significantly higher rates of coronary calcium (68%) and total plaque (77%) compared with less active athletes who exercised under 1,000 MET-minutes per week.
The second phase, MARC-2, was even more revealing. This longitudinal follow-up re-scanned 287 to 289 athletes from the original cohort approximately 6.3 years later, allowing researchers to actually watch atherosclerosis develop and progress in real time.6 The most important finding: it was not how much the athletes exercised that drove plaque progression — it was how hard they pushed.
Specifically, the proportion of exercise time performed at very vigorous intensity (≥9 METs — think hard interval running, all-out cycling efforts, or demanding rowing) was independently associated with greater acceleration of calcium scores and plaque expansion. Meanwhile, vigorous exercise at a more moderate intensity (6–9 METs — a brisk run you can sustain for an hour) was actually associated with less CAC progression. Total hours logged per week, stripped of intensity information, showed no independent association with plaque progression at all.
| The Intensity Question
This finding has real practical implications. The cardiovascular risk signal in endurance athletes appears to come specifically from the extreme end of the intensity spectrum — repeated near-maximal efforts sustained over years and decades — rather than from the accumulated volume of moderate training. This does not mean you should stop exercising hard. It means that understanding the nuance matters for how we screen and monitor aging athletes. |
The UK Masters Athlete Study: A Closer Look at Plaque Composition
In 2017, a landmark paper published in the journal Circulation added crucial nuance to the picture.7 Researchers in the United Kingdom compared 152 masters endurance athletes — predominantly runners, average age 54, with a median of 13 completed marathons — to 92 controls who were matched for age, sex, and cardiovascular risk profiles. Both groups had similarly low Framingham Risk Scores. The difference was that the athletes had been training intensely for an average of 31 years.
Male athletes had significantly more coronary plaque than controls (44.3% vs. 22.2%). Severe calcification (calcium scores ≥300 Agatston units) appeared only in the athlete group. So did significant narrowing of the coronary arteries — cases where plaque was blocking 50% or more of the vessel’s interior — which appeared in 7.5% of male athletes and 0% of the control group.
When researchers looked at the type of plaques the athletes carried, however, they found something initially reassuring: 72.7% of the plaques in male athletes were purely calcified — the dense, stable kind — while the sedentary controls had predominantly mixed-morphology plaques, which are generally considered more dangerous because they are more prone to rupturing and triggering a heart attack.
Fourteen percent of the male athletes (15 men) showed signs of myocardial scarring on cardiac MRI — a pattern consistent with previous, often silent, myocardial infarctions. Interestingly, only 3 of these 7 men with infarct-pattern changes had significant coronary blockages in the relevant artery, suggesting that some cardiac injury in extreme endurance athletes may come from mechanisms other than classic plaque rupture — including temporary coronary artery spasm, demand ischemia at peak exertion, or microvascular damage.7
The Master@Heart Study: The Definitive Challenge to the ‘Stable Plaque’ Theory
The reassuring conclusion from the UK Masters study — that athlete plaques are predominantly stable, calcified lesions — held sway for several years. Then, in 2023, a multicenter Belgian trial called Master@Heart published its results in the European Heart Journal, and the picture became more complicated.8
Master@Heart was the first study designed specifically to measure the absolute prevalence of all plaque types in athletes versus non-athletes, rather than just the relative proportion of plaque types in those who already had disease. This distinction matters enormously, because if athletes have far more total plaque than non-athletes, even a similar proportion of non-calcified plaque translates into a much higher absolute number of dangerous lesions.
The study enrolled 191 lifelong endurance athletes, 191 athletes who had taken up endurance sports after age 30, and 176 healthy but non-athletic controls. Critically, all participants were male and all had been screened to exclude anyone with traditional cardiovascular risk factors like high blood pressure, diabetes, or high cholesterol — isolating the physiological effects of exercise itself.
The results were unambiguous. Lifelong athletes had more of every type of plaque: more calcified plaques, more non-calcified plaques, and more mixed plaques — all statistically significant after controlling for confounding variables. They were also more likely to have plaques in the proximal (near the origin) segments of their coronary arteries, where blockages carry the greatest risk of causing large heart attacks.
The odds of a lifelong athlete having a non-calcified plaque in a proximal coronary segment was 2.80 times higher than for a non-athletic but healthy control.8 The study definitively showed that athlete atherosclerosis is not merely an acceleration of stable calcification — it is a full-spectrum progression of the disease process, including the higher-risk plaque types.
There was one important qualification, however: vulnerable plaques — defined by the presence of two or more high-risk imaging features — were actually less common in lifelong athletes than in controls (with an odds ratio of just 0.11). This suggests that while athletes accumulate more plaque overall, the structural characteristics of those plaques may still be more resistant to the kind of sudden rupture that triggers a heart attack. We will return to this mechanistic question later in the article.
| The Bottom Line from the Research: Male masters athletes carry more total coronary plaque than sedentary or moderately active people — including more non-calcified plaque in dangerous locations. But the plaques appear to be structurally more stable, and their overall cardiovascular mortality remains significantly lower. This is the paradox. |
What About Women?
Here is a finding that doesn’t receive nearly enough attention: the athlete calcification paradox appears to be almost entirely a male phenomenon.
A 2024 study published in Circulation by Papatheodorou and colleagues specifically examined coronary atherosclerosis in 196 highly competitive female masters athletes — women with an average age of 54, training about 8 hours per week, and a median of 33 years of endurance sports behind them — compared with 59 matched female controls.9
The findings were essentially the opposite of what we see in men. Female athletes actually had lower rates of elevated coronary calcium scores relative to their age and demographic compared with controls. They had fewer mixed-morphology plaques. None of the female athletes — and none of the controls — had obstructive coronary disease (stenosis blocking more than 50% of a vessel). Most importantly, the amount, duration, or intensity of exercise had no measurable relationship with calcium burden in women at all.
In female athletes, plaque — when it existed — was driven by traditional risk factors: advancing age, elevated systolic blood pressure, and low HDL cholesterol. Exercise was essentially neutral in its impact on arterial calcification in this population.
Researchers believe this sex difference is likely mediated by several protective biological mechanisms unique to women, including the prolonged endothelial-protective effects of estrogen prior to menopause, differences in hemodynamic responses to high cardiac output, and potentially lower inflammatory reactivity to the mechanical stress of high-intensity exercise.9 The practical implication is significant: the watchfulness we apply to aging male endurance athletes may not need to be applied equally to their female counterparts, at least regarding coronary calcification.
Exercise, Mortality, and the Dose-Response Question
To understand the athlete calcification paradox properly, we need to step back and look at the broader relationship between exercise and mortality — because it’s more nuanced than most people realize.
The relationship between physical activity and longevity follows what researchers describe as a reverse J-shaped curve.10 As you move from completely sedentary to lightly active to moderately active, your risk of dying from any cause drops dramatically. The largest gains in survival come at the low end of the activity spectrum — going from doing nothing to doing something is the single most powerful intervention available for longevity.
As you continue increasing your activity level beyond moderate, you continue to see mortality benefits, but they grow progressively smaller. Large analyses of prospective cohort studies suggest that people exercising two to four times the minimum recommended guidelines see an additional 2% to 13% reduction in mortality risk compared with those just meeting the guidelines.11 Beyond that, at volumes exceeding 3,000 MET-minutes per week — equivalent to running more than 4 miles every single day at a vigorous pace — the mortality benefit curve flattens out entirely.
Crucially, even at those extreme volumes, mortality risk does not increase. The data consistently shows that very high exercise volumes are associated with greater coronary plaque accumulation in men, but not with greater cardiovascular death rates.10 This is precisely the paradox: more structural disease burden, but no increase in mortality — and, in fact, a sustained protective effect compared with sedentary individuals.
| How Much Exercise Is Enough?
Current guidelines recommend 150–300 minutes of moderate physical activity per week, or 75–150 minutes of vigorous activity. Research confirms that this range delivers the largest survival benefits relative to effort invested. Going beyond this provides smaller additional gains but does not appear to increase mortality risk — even in the presence of coronary calcification. |
The Cooper Center Longitudinal Study: Athletes With Calcium Still Do Better
Perhaps the most directly relevant data for understanding the clinical significance of coronary calcium in athletes comes from the Cooper Center Longitudinal Study, which followed 21,758 generally healthy men without known cardiovascular disease for an average of 10.4 years.12
Among men who exercised the most — accumulating ≥3,000 MET-minutes per week — and who had low coronary calcium scores (under 100 Agatston units), their mortality risk was approximately half that of the least active men with comparably low calcium scores (hazard ratio 0.52; 95% confidence interval 0.29–0.91). Exercise dramatically reduced their mortality risk.
Among men who exercised the most but who had high coronary calcium scores (100 AU or above), there was no significant increase in all-cause mortality compared with the least active men who also had high calcium scores (hazard ratio 0.77; 95% CI 0.52–1.15). Despite carrying more structural disease, the highly active men were no more likely to die — and trended toward lower mortality, though the difference was not statistically significant.
Meanwhile, among the least active men, those with high calcium scores were nearly twice as likely to die from cardiovascular disease compared with those with low calcium scores (hazard ratio 1.93; 95% CI 1.34–2.78). This shows the flip side of the coin: inactivity dramatically amplifies the mortality risk associated with coronary calcification. Exercise, by contrast, appears to substantially blunt it.
The takeaway is powerful: finding coronary calcium in an athlete does not carry the same prognosis as finding it in someone who has been sitting on a couch. The calcification paradox is real — and it is clinically meaningful.
Why Does This Happen? The Biology Behind the Paradox
At this point, a reasonable question is: how does a lifestyle associated with extraordinary cardiovascular health produce more coronary plaque? And how does the same lifestyle protect against the lethal consequences of that plaque? The answers lie in four interconnected biological mechanisms.
-
Mechanical Stress on the Coronary Arteries
When you exercise at high intensity, your heart can pump five to six times more blood per minute than at rest. All of that blood flow has to travel through the coronary arteries — the vessels that supply the heart itself — creating substantial increases in local pressure, shear stress, and pulsatile force on arterial walls.3
This is especially pronounced at the branch points and curves of the coronary tree, where blood flow naturally becomes turbulent. The proximal left anterior descending artery — the vessel colloquially known as the “widow-maker” when blocked — is particularly susceptible to this turbulent flow pattern during sustained high-intensity exercise.13
Repeated over years and decades, this mechanical stress causes microdamage to the delicate inner lining of the arteries. The body responds to this damage the way it responds to any injury: it sends repair signals. Vascular smooth muscle cells migrate into the arterial wall and begin a process of mineral deposition — essentially hardening the damaged area. The calcium that shows up on a CAC scan is, in part, the accumulated record of this mechanical repair process.3
This is why athlete calcification is mechanistically different from the calcification seen in sedentary individuals with metabolic syndrome. In the latter, plaque forms primarily because of lipid accumulation and chronic low-grade inflammation driven by high cholesterol, insulin resistance, and other metabolic dysfunction. In the athlete, calcification is more directly a response to repeated hemodynamic stress — a localized structural reinforcement of the arterial wall rather than a sign of systemic metabolic failure.
-
The Parathyroid Hormone Connection
Every time you engage in intense endurance exercise, your body briefly elevates levels of parathyroid hormone (PTH) — the primary regulator of calcium in your blood and bones.3 The concentration and duration of this PTH spike correlates directly with how long and how hard you exercise.
PTH’s job is to keep blood calcium levels stable. When it rises, it mobilizes calcium from bone mineral stores into the bloodstream. Over decades of training, these repeated PTH pulses provide the raw mineral material for arterial calcification — the calcium mobilized from bone becomes available to deposit at the sites of endothelial damage described above.
This helps explain why the intensity of exercise matters more than volume: a hard interval session that spikes PTH substantially may drive more calcification than three hours of gentle jogging. And it helps explain why a 60-year-old who has been doing high-intensity training since his twenties carries more calcium in his coronary arteries than a man of the same age who has been running similar mileage at a more moderate effort.
-
The Fortified Fibrous Cap — Nature’s Armor
Now for the protective side of the equation. If athletes accumulate more plaque — including more non-calcified plaque — why aren’t they having more heart attacks?
A heart attack is not simply the result of having plaque in your coronary arteries. It requires that plaque to rupture — specifically, that the thin membrane covering the lipid core of the plaque (called the fibrous cap) tears, exposing the interior of the plaque to the bloodstream and triggering an explosive blood clot that blocks the artery.
In athletes, the same mechanical stress that drives plaque formation also appears to drive the construction of an unusually thick, dense fibrous cap. The intense, repetitive loading forces the vascular smooth muscle cells in the arterial wall to lay down a more robust structural matrix around the plaque — essentially building armor around the vulnerable core.14 This reinforced cap may be what protects athletes from the rupture events that would be far more likely in the same amount of plaque in a sedentary individual.
The Master@Heart data directly supports this. Despite carrying more total plaque volume, lifelong athletes were significantly less likely to have vulnerable plaques — those with multiple high-risk imaging features associated with imminent rupture — than non-athletic controls (odds ratio 0.11).8 More plaque overall, but more stable plaque. The biology is working against disease expression even while it is building disease structure.
-
Enlarged Arteries and the Collateral Circulation Advantage
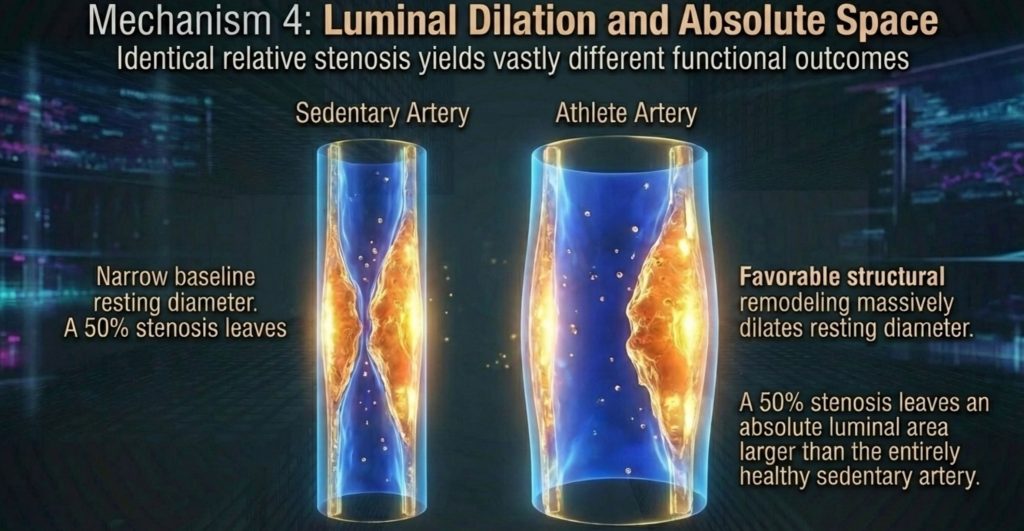
Lifelong endurance training reshapes the coronary arteries themselves, not just the plaques within them. Athletes’ coronary arteries tend to be significantly larger in diameter than those of sedentary individuals.8 This matters enormously when calculating the hemodynamic consequences of a given plaque.
A plaque that occupies 50% of the cross-sectional area of a normal-diameter artery may still allow adequate blood flow through the remaining half. A plaque that occupies 50% of a significantly dilated artery leaves an even larger absolute residual lumen — often enough that the blockage causes no meaningful reduction in blood flow, even during exercise. The mathematical 50% stenosis, measured by imaging, does not translate into functional ischemia the way it would in an artery of standard size.
Beyond this, endurance athletes develop rich networks of coronary collateral circulation — microscopic vessels that form over years of training to provide alternative routes for blood flow around partial blockages.8 If a vulnerable segment of a coronary artery does eventually occlude, this pre-existing collateral network can provide immediate backup perfusion, dramatically limiting the size of any resulting myocardial infarction and reducing the likelihood of fatal arrhythmia. It is, in effect, a biological redundancy system built by decades of cardiovascular demand.
What This Means If You’re an Aging Athlete — Or Treating One
The research we’ve described has significant practical implications, both for athletes themselves and for the physicians who care for them. The evidence is now clear enough that a new clinical framework is needed — and the American cardiology community has responded with updated guidance.
Standard Risk Calculators Will Miss You
One of the most consistent findings across every cohort we’ve discussed is that traditional cardiovascular risk tools — the Framingham Risk Score, the Pooled Cohort Equations, and similar calculators — substantially underestimate the true plaque burden in masters athletes.4 These tools were built on populations dominated by sedentary individuals, and they have no mechanism for capturing the mechanical, exercise-driven calcification described above.
A 58-year-old male masters athlete with ideal cholesterol, ideal blood pressure, and no smoking history will score as “low risk” on virtually every standard calculator — even if he has a CAC score of 400 and non-calcified plaques in his proximal LAD. Relying solely on these tools to make clinical decisions about aging athletes is no longer adequate.
Coronary Artery Calcium Scoring Changes the Picture
A CAC scan — a low-dose, non-contrast CT scan that takes about 10 minutes and involves no injections or dye — provides a direct measurement of calcified plaque burden and is highly effective at reclassifying risk in athletes.3 When a masters athlete who appears low-risk by traditional scoring turns out to have a high calcium score, it changes the management conversation: it identifies a person who may benefit from statin therapy, closer monitoring, and more detailed functional testing.
However, CAC scanning has a significant limitation in this population: it only detects calcified plaque. As the Master@Heart data shows, athletes also carry more non-calcified plaque — and CAC scoring is blind to it. A normal CAC score does not guarantee a clean bill of arterial health in a lifelong endurance athlete the way it might in a sedentary person.
When to Consider a Coronary CT Angiogram
For athletes who are symptomatic — who experience chest discomfort, unexplained shortness of breath, exercise-induced arrhythmias, or unusual drops in exercise capacity — a coronary CT angiogram (CCTA) provides far more information than a calcium score alone.8 Unlike a CAC scan, CCTA can visualize both calcified and non-calcified plaque, quantify the degree of narrowing in each vessel, and identify high-risk plaque features. When combined with cardiopulmonary exercise testing, which measures actual cardiac function during physiological stress, it gives clinicians a comprehensive picture of both anatomical disease and functional impact.
The decision to pursue CCTA in an asymptomatic athlete is more nuanced and should involve a shared conversation between the athlete and their physician about symptoms, risk factors, family history, and exercise goals.
The 2025 AHA/ACC Guidelines: Shared Decision-Making Over Restriction
In 2025, the American Heart Association and American College of Cardiology published a comprehensive scientific statement on cardiovascular considerations for competitive sports participation in athletes with cardiovascular abnormalities.15 The document represents a significant philosophical shift in how the cardiology community approaches this population.
Where earlier eras of sports medicine leaned toward caution — disqualifying athletes with abnormal findings from competitive participation — the 2025 statement explicitly endorses a framework of shared decision-making. Rather than physicians unilaterally restricting athletic activity because of imaging findings, the guidelines call for open communication between clinician and athlete about the available evidence, the existing uncertainties, and the athlete’s own values and priorities.
Key 2025 AHA/ACC Guideline Principles for Masters Athletes With Coronary Disease
| Domain | What the 2025 Guidelines Say |
| Decision-Making | Shared decision-making is the standard of care — not physician-directed restriction. The athlete’s values, quality of life, and the proven benefits of exercise must be weighed alongside anatomical findings. |
| Continuing Exercise | Elevated CAC does not automatically mean an athlete must stop competing. If high-risk ischemia and dangerous arrhythmias are ruled out through stress testing, continued participation is actively supported. |
| Medical Treatment | Athletic fitness does not reduce the indication for evidence-based medical therapy. Significant plaque burden warrants consideration of statin therapy and rigorous blood pressure management. |
| Emergency Preparedness | Sudden cardiac arrest can occur even in well-screened athletes. All athletic environments should have AEDs and trained responders — this is non-negotiable. |
AED = automated external defibrillator; AHA/ACC = American Heart Association/American College of Cardiology; CAC = coronary artery calcium. Adapted from the 2025 AHA/ACC Scientific Statement.15
The guidelines also emphasize that finding calcification in an athlete’s arteries is never a reason to abandon medical therapy. Statin medications and blood pressure management remain the cornerstone of secondary prevention regardless of fitness level. High cardiovascular fitness does not confer immunity to pharmacological benefit.
Putting It All Together: What We Know and What Remains Uncertain
The story of coronary artery disease in masters athletes is, in many ways, a story about biological complexity resisting simple narratives. The exercise-is-always-good story turns out to be incomplete at extreme doses and over extreme time horizons. But the exercise-is-dangerous story is equally wrong. The truth is more interesting than either.
Here is a fair summary of what we can say with confidence, based on the current evidence:
What we know: Male masters athletes accumulate significantly more coronary plaque than sedentary or moderately active people of the same age and risk profile — including non-calcified plaque in dangerous proximal locations. This plaque accumulation is driven by the mechanical stress of high-intensity exercise and the calcium-mobilizing effects of repeated parathyroid hormone elevation. Exercise intensity — not total volume — appears to be the primary driver of CAC progression. Despite this, all-cause and cardiovascular mortality remain substantially lower in highly active populations, even when they carry elevated coronary calcium. Female masters athletes are largely protected from this paradox; their coronary plaque burden is driven by traditional risk factors, not by exercise. Standard risk calculators dramatically underestimate plaque burden in athletes. And the 2025 AHA/ACC guidelines call for individualized, shared decision-making rather than blanket restriction.
What remains uncertain: We do not yet have long-term clinical outcomes data specifically from high-volume athlete cohorts. We do not fully understand why some athletes develop significant non-calcified plaque while others with similar training histories do not. We do not know the precise threshold of exercise intensity or duration beyond which the structural risks outweigh the systemic benefits — if such a threshold exists at the population level at all. And we do not have clear guidance on when, or whether, the discovery of subclinical coronary disease in an asymptomatic athlete should change their training behavior.
| The most important thing to understand is this: if you are an aging endurance athlete, discovering coronary calcium in your arteries does not mean your years of training have hurt you. The evidence strongly suggests the opposite — that your activity has dramatically reduced your risk of dying from the disease, even as the mechanical demands of training have left their mark on your arterial walls. But it does mean that you deserve informed, individualized cardiovascular care — not dismissal, and not panic. |
Conclusion: The Enduring Power of Movement
The athlete calcification paradox is one of the most fascinating puzzles in contemporary cardiology, and it is far from fully solved. But its broad contours are now clear enough to draw some confident conclusions.
Decades of high-intensity endurance exercise do leave a structural signature on the coronary arteries of aging male athletes — more plaque, including some plaque types traditionally considered high-risk, in anatomically vulnerable locations. This is not a myth or an artifact of measurement. It is a real biological phenomenon driven by real physiological mechanisms: hemodynamic stress, parathyroid hormone, vascular smooth muscle cell behavior, and the body’s attempt to repair repeated arterial injury.
And yet — the same decades of exercise that create these structural changes simultaneously build an extraordinary suite of cardiovascular protections: thicker, more stable fibrous caps that resist rupture; enlarged arteries that accommodate plaque without the same functional consequences; supranormal coronary flow reserve; and rich collateral networks that provide redundancy against acute occlusion. The result is a cardiovascular system that is simultaneously more diseased and more protected than its sedentary counterpart.
The ultimate measurement that matters — survival — comes down clearly in favor of the athlete. High levels of physical activity are protective against cardiovascular mortality even in the presence of significant coronary calcification. Inactivity, by contrast, amplifies the lethal potential of the same anatomical findings dramatically.
None of this means you should ignore coronary findings in aging athletes. It means you should interpret them correctly — in the full context of the physiology, with the right tools, and in partnership between informed clinicians and informed patients. That is precisely what the 2025 AHA/ACC guidelines are now calling for. And it is, ultimately, what good medicine has always looked like: meeting complexity with nuance, and evidence with humility.
Medical Disclaimer
The information presented in this article is for educational purposes only and is based on peer-reviewed scientific literature. It does not constitute medical advice, a diagnosis, or a treatment recommendation. Always consult a qualified healthcare provider before making changes to your exercise routine, pursuing cardiovascular testing, or starting or stopping any medication. Individual risk profiles vary widely, and no general population-level finding should be applied without professional clinical evaluation.
References
All sources cited in this article are peer-reviewed publications. References are provided for readers who wish to explore the primary literature.
- Bassler TJ. More on immunity to atherosclerosis in marathon runners. N Engl J Med. 1978;299(4):201. doi:10.1056/NEJM197807272990417
- Waller BF, Roberts WC. Sudden death while running in conditioned runners aged 40 years or over. Am J Cardiol. 1980;45(6):1292-1300. doi:10.1016/0002-9149(80)90491-9
- Hsu JJ, Tintut Y, Demer LL. Paradox of Exercise and Coronary Artery Calcification: Potential Underlying Mechanisms. Circ Res. 2025;137(2):335-349. doi:10.1161/CIRCRESAHA.125.326011
- Möhlenkamp S, Lehmann N, Breuckmann F, et al. Running: the risk of coronary events : Prevalence and prognostic relevance of coronary atherosclerosis in marathon runners. Eur Heart J. 2008;29(15):1903-1910. doi:10.1093/eurheartj/ehn163
- Aengevaeren VL, Mosterd A, Braber TL, et al. Relationship Between Lifelong Exercise Volume and Coronary Atherosclerosis in Athletes. Circulation. 2017;136(2):138-148. doi:10.1161/CIRCULATIONAHA.117.027834
- Aengevaeren VL, Mosterd A, Bakker EA, et al. Exercise Volume Versus Intensity and the Progression of Coronary Atherosclerosis in Middle-Aged and Older Athletes: Findings From the MARC-2 Study. Circulation. 2023;147(13):993-1003. doi:10.1161/CIRCULATIONAHA.122.061173
- Merghani A, Maestrini V, Rosmini S, et al. Prevalence of Subclinical Coronary Artery Disease in Masters Endurance Athletes With a Low Atherosclerotic Risk Profile. Circulation. 2017;136(2):126-137. doi:10.1161/CIRCULATIONAHA.116.026964
- De Bosscher R, Dausin C, Claus P, et al. Lifelong endurance exercise and its relation with coronary atherosclerosis. Eur Heart J. 2023;44(26):2388-2399. doi:10.1093/eurheartj/ehad152
- Papatheodorou E, Aengevaeren VL, Eijsvogels TMH, et al. Prevalence of Coronary Atherosclerosis in Female Masters Endurance Athletes. Circulation. 2024;150(18):1478-1480. doi:10.1161/CIRCULATIONAHA.124.069484
- Eijsvogels TM, Molossi S, Lee DC, Emery MS, Thompson PD. Exercise at the Extremes: The Amount of Exercise to Reduce Cardiovascular Events. J Am Coll Cardiol. 2016;67(3):316-329. doi:10.1016/j.jacc.2015.11.034
- Lee DH, Rezende LFM, Joh HK, et al. Long-Term Leisure-Time Physical Activity Intensity and All-Cause and Cause-Specific Mortality: A Prospective Cohort of US Adults. Circulation. 2022;146(7):523-534. doi:10.1161/CIRCULATIONAHA.121.058162
- DeFina LF, Radford NB, Barlow CE, et al. Association of All-Cause and Cardiovascular Mortality With High Levels of Physical Activity and Concurrent Coronary Artery Calcification. JAMA Cardiol. 2019;4(2):174-181. doi:10.1001/jamacardio.2018.4628
- Wasilewski J, Czaja-Ziółkowska MZ, Gąsior M. The site-specific distribution of atheromatous plaques in the coronary arteries. Postepy Kardiol Interwencyjnej. 2023;19(3):195-201. doi:10.5114/aic.2023.131471
- Libby P, Pasterkamp G, Crea F, Jang IK. Reassessing the Mechanisms of Acute Coronary Syndromes. Circ Res. 2019;124(1):150-160. doi:10.1161/CIRCRESAHA.118.311098
- Kim JH, Baggish AL, Levine BD, et al. Clinical Considerations for Competitive Sports Participation for Athletes With Cardiovascular Abnormalities: A Scientific Statement From the American Heart Association and American College of Cardiology. Circulation. 2025;151(11):e716-e761. doi:10.1161/CIR.0000000000001297