1. The “Normal” Lab Result Mystery
Imagine a man named Mark. Mark is 48 years old, and lately, he feels like a shell of his former self. Every day around 3:00 PM, he hits a “wall” of exhaustion that feels like swimming through deep mud. His “brain fog” makes it hard to focus at work, and he has completely lost the “spark” for his hobbies and his relationships. He is tired, moody, and just wants to nap.
Hoping for an answer, Mark goes to his doctor. The doctor runs a standard blood test and looks at a single number: “Total Testosterone.” The doctor smiles and says, “Good news, Mark! Your score is 500. That is perfectly normal for a man your age. You are just getting older.”
Mark leaves the office feeling even worse. If his numbers are “normal,” why does he feel so broken? The truth is that Mark’s “Total Testosterone” score is lying to him. In the world of men’s health, that big number on your lab report is often just biochemical noise. It is a confusing sound that hides the real music of your health. It tells you how much hormone is in your blood, but it does not tell you how much your body can actually use.
A major new clinical review has revealed that our old way of looking at men’s hormones is missing the point. To understand why, think of your testosterone like a bank account. Your Total Testosterone is the total balance shown on the screen. However, for many men, most of that money is “frozen” in a locked vault. You can see it, but you can’t spend it. If you can’t spend the money, you can’t buy a sandwich, and you will stay hungry. In the same way, if your body can’t “spend” its testosterone, you will feel all the symptoms of being low, no matter what the bank balance says.
This post will reveal the five hidden truths about men’s health that explain why “normal” isn’t always what it seems.
2. The “Locked Vault” Problem: Total vs. Bioavailable Testosterone
To truly understand your health, you have to look past that “Total” number. Your body carries testosterone in three different “buckets.” How it is stored determines whether or not it can actually help you feel better.
- Free Testosterone: This is like the cash in your pocket. It is floating freely in your blood, ready to be used by your brain, muscles, and bones at a moment’s notice. It can “diffuse,” or soak, right into your cells.
- Albumin-bound Testosterone: This is like money in a piggy bank. It is attached to a protein called albumin, but it is a “loose” connection. Your body can break that piggy bank open very easily when it needs extra help during its trip through your blood vessels.
- SHBG-bound Testosterone: This is the money in the locked vault. It is tightly gripped by a protein called Sex Hormone-Binding Globulin (SHBG). This protein is like an overprotective bodyguard that refuses to let go.
When experts talk about Bioavailable Testosterone, they are talking about the Free and Albumin-bound parts together. This is the only number that truly matters for how a man feels. Why? Because the part stuck to the SHBG protein is basically invisible to your cells. As the new clinical review explains:
“The portion that is tightly bound to SHBG is, for practical purposes, biologically inert.”
“Biologically inert” is a fancy way of saying “dead weight.” It is in your blood, but it isn’t doing any work. If your SHBG levels are high, they act like a giant sponge, soaking up all your testosterone and locking it away. You could have a “high” total score of 800, but if your bodyguard (SHBG) is too strong, you might have almost zero usable testosterone. This is the “Locked Vault” problem, and it is why Mark feels terrible even though his doctor said he was fine.
3. SHBG: The Bodyguard That Won’t Let Go
If bioavailable testosterone is the “spendable cash,” then SHBG is the gatekeeper deciding how much you get to keep. SHBG is a protein made by your liver factory. Its job is to regulate how much hormone is active in your system.
Sometimes, this factory goes into overdrive. When SHBG levels rise, your usable testosterone levels crash. This creates a very strange situation: a man can look “healthy” and lean on the outside, but because his SHBG is too high, his body is actually starving for the hormone.
According to the latest research, here is what causes the “bodyguard” to change its behavior:
What makes SHBG go UP (locking away your testosterone):
- Aging: Starting in your 30s, SHBG levels naturally creep up by about 1% every year. It’s like the vault door gets harder to open as the years pass.
- Hyperthyroidism: An overactive thyroid acts like a boss yelling at the liver to pump out more SHBG bodyguards.
- Liver Issues: Certain types of liver disease, like cirrhosis, can cause SHBG to spike.
What makes SHBG go DOWN (releasing more testosterone):
- Obesity: Carrying extra weight, especially around the belly, sends signals to the liver to make less SHBG.
- High Insulin and Sugar: This is a big discovery. When you eat a lot of sugar or high-fructose corn syrup, it actually flips a “factory switch” in your liver. It shuts down the gene that makes SHBG. This is why men with diabetes often have very low “Total T”—their bodyguard is gone, so the testosterone doesn’t stay in the blood long.
- Steroids: Using artificial muscle-building drugs can crush SHBG levels.
It is counter-intuitive, but a lean, fit older man might actually have less usable testosterone than a man who is slightly overweight. This is because the lean man’s SHBG might be so high that it has locked all his hormones in the vault, while the other man’s lower SHBG is letting more hormone flow freely.
4. The Libido “North Star”: Why Desire is the Only Reliable Signal
When men talk to doctors about hormones, they often focus on “Erectile Dysfunction” (ED). But the latest science shows that ED is actually a very poor way to measure your hormones.
ED is usually a “plumbing” problem. It is about blood vessels, heart health, or even just being stressed out. It’s like having a kink in a garden hose. A man can have perfect hormone levels and still have ED if his blood vessels aren’t healthy. Conversely, a man can have very low hormones and still be able to perform.
If you want to know if your hormones are low, you have to look at your “North Star”: Sexual Desire (Libido).
Low desire is a direct signal from the brain. It is the most specific sign that your bioavailable testosterone has dropped. Researchers have found what they call a “graded relationship” between the two. This means that as your usable testosterone goes down, your desire goes down right along with it, step-by-step. They move together like a shadow. This isn’t true for the “Total” score, which is why your libido is a much better guide than a standard lab test.
“There is a graded relationship between bioavailable testosterone and libido that holds across the physiological range.”
What does this feel like? It isn’t just about “not being able to perform.” Men with low bioavailable testosterone describe a loss of “spontaneous erotic thought.” They notice they simply don’t think about romance unprompted anymore. The “internal drive” or the “spark” is gone. If you still have the desire but have trouble with performance, it might be a blood pressure or heart issue. But if the “wanting” is gone, that is a hormone signal you shouldn’t ignore.
5. The Heart Attack Myth: What the TRAVERSE Trial Actually Found
For a long time, many men—and even many doctors—were afraid of testosterone therapy. Back in 2013 and 2014, a few small studies suggested that taking testosterone might cause heart attacks or strokes. These studies were not very high quality, but they caused a lot of fear. In fact, the FDA even put a “boxed warning”—the scariest kind of label—on testosterone products.
That has all changed thanks to a massive, gold-standard study called the TRAVERSE trial.
This study followed 5,200 men for about two years. These weren’t just healthy young men; they were men between 45 and 80 who already had heart disease or a very high risk for it. Half the men took testosterone, and half took a “placebo” (a dummy treatment).
The results were clear: there was no difference in heart attacks or strokes between the two groups. Testosterone did not make heart problems worse. Because of this high-quality evidence, the FDA officially removed the “boxed warning” about heart risks in 2025.
However, a good health journalist must mention the new nuances. While the heart attack fear was lowered, the FDA strengthened warnings about blood pressure. Testosterone can make blood pressure go up in some men, so it must be watched.
Doctors also watch for Erythrocytosis, which is just a fancy word for “thick blood.” Testosterone tells your body to make more red blood cells. If your blood gets too thick—like syrup—it can be harder for your heart to pump it. Doctors look for a “red line” of 54% hematocrit. If your blood gets thicker than that, it doesn’t mean you have to stop therapy, but it usually means your doctor needs to adjust your dose to keep things moving smoothly.
6. Keeping the Factory Open: The Role of hCG
When a man starts standard testosterone therapy, something happens that most people don’t expect: his body’s own “testosterone factory” (the testicles) shuts down.
Think of it like a business. Your brain is the manager. When you start “importing” testosterone from a pharmacy, the brain sees all this hormone and thinks, “Well, the job is already done! We don’t need to work anymore.” The manager then “lays off the workers” and closes the factory doors. This can lead to testicular atrophy (the shrinking of the testicles) and a loss of fertility (the ability to have children).
This is where a helper called hCG comes in. hCG acts like a government grant that keeps the workers paid even when you are importing goods from outside. It sends a “fake” signal to the factory that says, “Hey, keep the power grid on! Keep working!”
Using hCG alongside testosterone is vital for two types of men:
- Men who want to stay fertile: It keeps the “machinery” running so they can still have children.
- Men who want to avoid shrinking: It maintains the physical size and health of the factory.
It ensures that while you are getting the benefits of therapy, your body doesn’t “forget” how to do its own job.
7. Conclusion: A New Way to Look at Aging
We have been taught that “feeling old” is just a debt we have to pay. We are told that being tired, losing our drive, and feeling “blah” is just part of the deal. But the science tells a different story.
The main takeaway is simple: Don’t just settle for a “Total T” number. If you feel the symptoms—especially a loss of desire and drive—ask your doctor to look at the Bioavailable fraction. Look at your SHBG levels. Check your liver health and your insulin levels, because they are the “hidden hands” controlling your hormones.
Stop looking at the total balance in the bank and start asking how much of that balance is actually in your pocket. How much of “feeling old” is really just a “locked vault” of hormones that could be opened with the right medical approach? By understanding the difference between “total” and “usable” hormones, you can stop guessing about your health and start getting the answers you deserve.
DEEP DIVE
Bioavailable Testosterone in Men: Pathophysiology, the Clinical Importance of Sexual Desire, Cardiovascular Implications, and Restorative Therapy with Testosterone and hCG
A clinical review for evidence-based preventive practice
Abstract
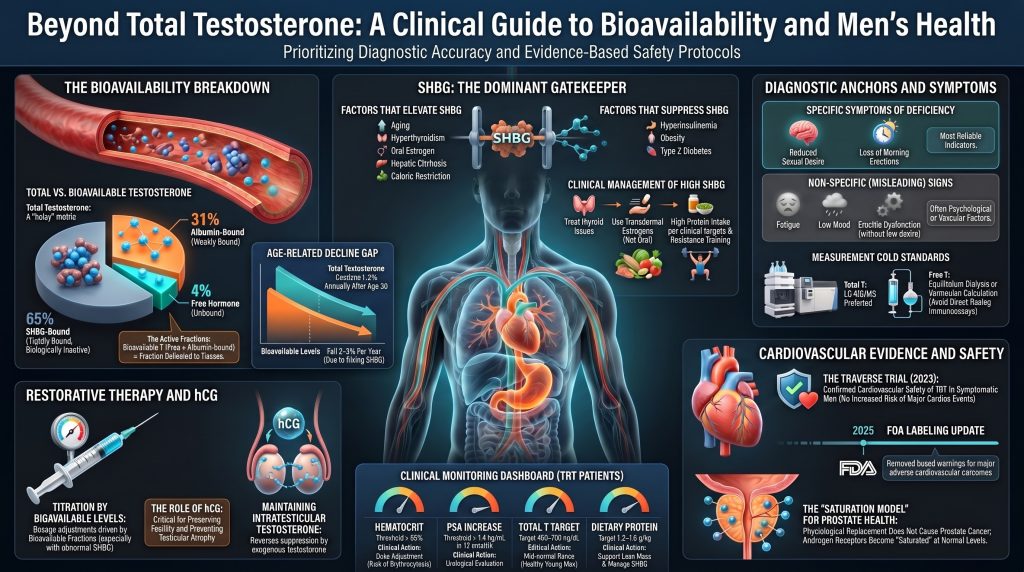
The clinical evaluation of male androgen status has moved well past the simple measurement of total serum testosterone. Total concentrations are routinely misleading because the majority of circulating testosterone is bound to plasma proteins of widely different affinities, with sex hormone-binding globulin (SHBG) acting as the principal regulator of how much hormone is actually delivered to tissue. This review consolidates the modern understanding of bioavailable testosterone, covering the biochemistry of compartmentalization, the mechanisms of age-related decline, the regulation of SHBG (including practical strategies to lower it when it is clinically elevated), the analytical methods that produce reliable measurements, and the clinical importance of reduced sexual desire as the most specific clinical signal of true androgen deficiency. The cardiovascular implications of low bioavailable testosterone are examined in detail, including the results of the TRAVERSE trial and the 2025 FDA labeling update. The therapeutic section addresses both testosterone replacement and the appropriate use of human chorionic gonadotropin (hCG), which preserves intratesticular testosterone production and spermatogenesis. The objective is a coherent, contemporary framework that clinicians can apply to male patients in practice without conflating biochemical noise with disease.
1. Introduction
A growing body of evidence has reframed how clinicians should think about testosterone. The hormone does not act on tissue in proportion to its total concentration in blood; it acts in proportion to the unbound fraction, plus a loosely bound fraction that releases from albumin during capillary transit. The portion that is tightly bound to SHBG is, for practical purposes, biologically inert. This is the substance of the free hormone hypothesis, and although it has been refined since its original formulation, its central claim has held up: bioavailable testosterone (the sum of the free and albumin-bound fractions) is the more meaningful biological variable.1-4
The clinical consequence is that two men with identical total testosterone concentrations can have very different androgen physiology if their SHBG concentrations differ. SHBG itself is regulated by hepatic processes that respond to insulin, thyroid hormones, sex steroids, inflammatory cytokines, nutritional status, and aging. As a result, common conditions such as obesity, type 2 diabetes, hyperthyroidism, hepatic dysfunction, and the aging process can decouple total testosterone from the hormone’s actual tissue activity.5-7
This review is organized around the questions a clinician needs to answer in practice: why testosterone falls with age, why SHBG rises, what matters most symptomatically (with sexual desire as the most diagnostically useful indicator), how the laboratory should measure these analytes, what the cardiovascular literature now says about both endogenous testosterone and replacement therapy, and how testosterone and hCG should be used to restore physiology safely.
2. Compartmentalization of Circulating Testosterone
2.1 Binding fractions and their biological consequences
In a healthy eugonadal man, roughly 44 to 65 percent of total testosterone is bound to SHBG, 30 to 50 percent is loosely bound to albumin, and 1 to 4 percent circulates as free hormone.1,2,3 SHBG is a glycoprotein homodimer of approximately 95 kDa, produced primarily by the liver. Its association constant for testosterone is on the order of 1 × 10⁹ per molar, sufficient to make the SHBG-bound fraction effectively unavailable during the brief transit through the capillary bed.1 Albumin, by contrast, is abundant but binds testosterone with a much lower affinity of approximately 3 × 10⁴ per molar.2,3 The albumin bond dissociates rapidly enough that the albumin-bound fraction can release into tissue during transit, which is why the sum of the free and albumin-bound fractions is termed bioavailable testosterone.2,4
The free hormone hypothesis holds that only the unbound molecule diffuses across the cell membrane to act on the intracellular androgen receptor.4 Recent in vivo data using highly sensitive assays have refined this picture, suggesting that the dissociation kinetics of SHBG and albumin allow for some bioavailability beyond the strictly free pool, particularly in tissues with slow capillary transit.2 The practical conclusion has not changed: total testosterone alone can mislead, and the clinically meaningful exposure is closer to the bioavailable fraction.
Table 1. Plasma testosterone fractions in adult men
| Fraction | Binding partner | Affinity (Ka, M⁻¹) | Share of total T | Clinical relevance |
| Free | None | Not applicable | 1–4% | Diffuses freely across cell membranes; active fraction |
| Albumin-bound | Albumin | ~3 × 10⁴ | 30–50% | Loosely bound; releases during capillary transit and contributes to tissue delivery |
| SHBG-bound | SHBG | ~1 × 10⁹ | 44–65% | Tightly bound; biologically inert under normal conditions |
| Bioavailable | Free + albumin-bound | Mixed | ~30–54% | Best index of tissue-level androgen exposure |
2.2 Why total testosterone is not enough
Because the SHBG-bound and albumin-bound fractions are governed by mass-action equilibria, a change in SHBG concentration shifts the partition between bound and free testosterone even if testicular production is unchanged. A doubling of SHBG, which is not unusual in the transition from young adulthood to the seventh decade, can normalize total testosterone while bioavailable testosterone has fallen substantially.2,9,13 This is the principal reason older men with normal-appearing total testosterone can present with clinically meaningful androgen-deficiency symptoms.
3. Why Testosterone Declines with Age
The age-related decline in androgen status in men is now well documented. The Baltimore Longitudinal Study of Aging followed healthy men over decades and reported a decline in total testosterone of about 1 to 2 percent per year after the third decade, with free and bioavailable testosterone declining more steeply at approximately 2 to 3 percent per year because SHBG rises in parallel.9 The Massachusetts Male Aging Study confirmed similar trajectories in a community-based cohort.10 The European Male Ageing Study (EMAS) further showed that in men with otherwise normal pituitary function the decline reflects a mixture of testicular and central mechanisms.11,15
3.1 Mechanisms of decline
Several pathways contribute simultaneously, which is why the syndrome of late-onset hypogonadism is heterogeneous.
Testicular aging. Leydig cell number declines, and the surviving Leydig cells show reduced steroidogenic capacity. Mitochondrial dysfunction, accumulation of lipofuscin, and impaired StAR-mediated cholesterol transport all contribute to lower per-cell testosterone output.13
Loss of hypothalamic-pituitary pulsatility. GnRH pulses become less frequent and lower in amplitude, LH pulses follow suit, and the LH amplitude required to drive testosterone secretion is reached less reliably. Veldhuis and colleagues showed that this loss of orderly pulsatility is itself an independent component of male reproductive aging.14
Body composition shift. Visceral adiposity expands across middle age, even in men who maintain a stable weight. Adipose tissue secretes inflammatory cytokines that suppress GnRH and pituitary gonadotropin secretion and contains aromatase, which converts testosterone to estradiol. The result is functional secondary hypogonadism layered on top of primary testicular aging.20,21,22
Rising SHBG. Hepatic SHBG synthesis increases by roughly one percent per year after the third decade in most men.2,9 Because SHBG sequesters testosterone, the same total testosterone yields a lower bioavailable fraction in older men. The Travison harmonization study established age-stratified reference ranges that account for this physiology.12
Secular trends. Travison and colleagues showed that population-level testosterone in American men has fallen across birth cohorts over recent decades independent of age, suggesting that environmental factors (likely including obesity prevalence, endocrine disruptors, and changes in physical activity) contribute on top of the intrinsic aging process.16
3.2 What aging does to symptoms
EMAS established that only a narrow set of symptoms tracks androgen status reliably in older men. Three sexual symptoms (poor morning erection, low sexual desire, and erectile dysfunction) together with a total testosterone below approximately 11 nmol/L (320 ng/dL) and a free testosterone below 220 pmol/L (64 pg/mL) define the most specific phenotype of late-onset hypogonadism.11 The other symptoms typically attributed to low testosterone, including fatigue, low mood, and reduced strength, are common in older men but track androgen status weakly.
4. SHBG: Regulation and Why It Matters
SHBG functions as the dominant gatekeeper of androgen and estrogen bioavailability. Its hepatic synthesis is regulated by HNF-4α, with permissive or suppressive input from insulin, thyroid hormones, sex steroids, inflammatory cytokines, and nutrient sensors.1,5,6,17 Importantly, low SHBG is now recognized as an independent biomarker of insulin resistance and incident type 2 diabetes, while high SHBG is a marker of hepatic and metabolic states that change the interpretation of any total testosterone value.18,19
4.1 Conditions that raise SHBG
Aging is the most common cause of elevated SHBG, with the steady upward drift described above. Hyperthyroidism raises SHBG through stimulation of HNF-4α and HNF-1α.17 Estrogen exposure (endogenous, from oral contraceptives, or from estrogen-replacement therapy) markedly induces hepatic SHBG synthesis, which is one mechanism by which oral but not transdermal estrogen lowers free testosterone in women.6 Hepatic cirrhosis frequently elevates SHBG (sometimes paradoxically so, given otherwise impaired hepatic synthetic function) due to altered hepatocyte metabolism and reduced clearance.24 Anorexia nervosa, severe caloric restriction, and chronic protein deprivation raise SHBG. Several anticonvulsants (phenytoin, carbamazepine) and some HIV medications induce SHBG via hepatic enzyme effects.6
4.2 Conditions that lower SHBG
Hyperinsulinemia is the single most clinically important cause of low SHBG in modern populations. Selva and colleagues demonstrated that monosaccharides (specifically fructose and glucose) suppress hepatic SHBG transcription through downregulation of HNF-4α, with hepatic lipogenesis as the proximate mediator.5 This explains why low SHBG so reliably accompanies metabolic syndrome, obesity, and type 2 diabetes, and why SHBG is now used as a biomarker in metabolic research.18,19,40 Other suppressors include hypothyroidism, exogenous androgens (including supraphysiologic testosterone, anabolic steroids, and danazol), glucocorticoid excess, growth hormone or IGF-1 excess, and progestins with androgenic activity.6
Table 2. Major modifiers of hepatic SHBG synthesis
| Raises SHBG | Lowers SHBG |
| Aging (steady rise from the third decade) | Hyperinsulinemia and insulin resistance |
| Hyperthyroidism | Obesity, especially visceral adiposity |
| Oral estrogen exposure (oral contraceptives, pregnancy) | Type 2 diabetes mellitus |
| Hepatic cirrhosis | Hypothyroidism |
| Caloric restriction and anorexia nervosa | Exogenous androgens and anabolic steroids |
| HIV infection (multifactorial) | Glucocorticoid excess (Cushing syndrome) |
| Anticonvulsants (phenytoin, carbamazepine) | Acromegaly (GH/IGF-1 excess) |
| Chronic inflammation (variable effect) | Nephrotic syndrome (urinary protein loss) |
4.3 Lowering clinically elevated SHBG by non-pharmacologic means
When SHBG is high in a clinically relevant context (commonly in older men with a relatively lean body habitus, in subclinical hyperthyroidism, or in men on oral estrogen for any reason), the priority is to address the underlying driver. The strategies below have the strongest mechanistic support, although the evidence base for many is observational rather than randomized.
Resistance training and adequate dietary protein. Exercise-induced increases in lean mass, particularly through resistance training, are associated with modest reductions in SHBG, likely through improved insulin sensitivity at the muscle level. Adequate protein intake (1.2 to 1.6 g/kg body weight in older active men) supports muscle mass accrual and limits the catabolic state that itself raises SHBG.20,21
Carbohydrate quality and glycemic load. The effect of dietary modification on SHBG depends on which driver predominates and must be considered separately across patient phenotypes. In insulin-resistant men with low SHBG, improving insulin sensitivity (by reducing refined sugar, fructose-sweetened beverages, and high-glycemic-load foods) tends to raise SHBG toward normal. In undernourished or energy-deficient men with high SHBG from caloric restriction, adequate caloric repletion tends to lower SHBG. In men with high SHBG attributable to thyroid disease, oral estrogen, anticonvulsants, or liver disease, dietary modification alone is insufficient; the appropriate intervention targets the underlying driver.5,18
Treating subclinical hyperthyroidism. When TSH is suppressed and SHBG is high, addressing the thyroid abnormality often normalizes SHBG within a few months.17
Reducing or replacing oral estrogen. Patients on oral estrogen for any reason can be transitioned to transdermal estrogen, which bypasses the first-pass hepatic effect and produces little change in SHBG.6
Reviewing medications. Anticonvulsants and certain antiretrovirals may need substitution if hypogonadism is confirmed and SHBG is the principal driver.6
Addressing hepatic disease. In men with chronic liver disease where SHBG is paradoxically elevated, treatment of the hepatic process (alcohol cessation, antiviral therapy for hepatitis C, or weight loss for hepatic steatosis) is the appropriate intervention.24
Adequate caloric intake. SHBG rises sharply in undernutrition. Athletes and older men in negative energy balance can present with high SHBG that resolves with caloric repletion.6,15
Boron, zinc, and magnesium supplementation are sometimes recommended for SHBG modulation. The clinical evidence is limited and inconsistent, and none of these substitutes for addressing the upstream drivers above.
5. Conditions That Decouple Total from Bioavailable Testosterone
5.1 Obesity and functional secondary hypogonadism
Obesity, particularly visceral obesity, produces a pattern that has been termed male obesity-associated secondary hypogonadism (MOSH).22 The pattern is characterized by low total testosterone with inappropriately normal or low LH and FSH, indicating a central component. Three mechanisms operate simultaneously: hyperinsulinemia suppresses hepatic SHBG, lowering total testosterone without necessarily lowering free testosterone; visceral adipose tissue secretes IL-6, TNF-α, and leptin in patterns that suppress hypothalamic GnRH; and adipose aromatase converts testosterone to estradiol, providing additional negative feedback to the hypothalamus.20,22,23 In men with diabesity, more than 30 percent meet the biochemical criteria for hypogonadism in cross-sectional studies.23
Crucially, weight loss and improvement in insulin sensitivity often partially or wholly reverse this picture. Bariatric surgery can normalize testosterone in obese hypogonadal men, and even modest weight loss of 5 to 10 percent improves both total and free testosterone.20,21,22
5.2 Metabolic dysfunction-associated steatotic liver disease (MASLD)
Hepatic steatosis is a phenotypic expression of hepatic insulin resistance and is closely associated with low SHBG and low total testosterone.19,40 Free or bioavailable testosterone is the more meaningful measurement in these patients. The presence of steatohepatitis adds a second layer because progression to fibrosis and cirrhosis can paradoxically raise SHBG.24
5.3 Hepatic cirrhosis
Advanced cirrhosis produces a complex endocrinopathy. SHBG rises despite reduced hepatic synthetic function in many other domains; albumin falls; estradiol clearance decreases. Total testosterone may appear deceptively preserved while free testosterone has collapsed.24 Bioavailable measurement is mandatory in this population; otherwise the diagnosis is missed.
5.4 Nephrotic syndrome
Massive proteinuria can produce urinary loss of SHBG and albumin, lowering both. Total testosterone falls because the bound carrying capacity has been reduced; free testosterone may initially be preserved by HPG-axis compensation, but chronic catabolic stress and direct cytokine effects on Leydig cells eventually impair production.25
5.5 Hyperthyroidism and hypothyroidism
Hyperthyroidism raises SHBG through HNF-1α and HNF-4α induction, elevating total testosterone but with smaller effects on bioavailable testosterone.17 Hypothyroidism produces the opposite pattern. In any patient where total testosterone is interpreted clinically, thyroid status should be known.
6. Cardiovascular Implications of Bioavailable Testosterone
The cardiovascular relevance of testosterone status is the part of this field that has changed most over the last two decades. Older literature treated testosterone as cardiovascularly neutral or harmful; the current evidence supports a more nuanced view in which physiological levels of bioavailable testosterone are associated with a favorable cardiovascular phenotype, while both very low and supraphysiological levels carry risk.
6.1 Vascular biology of testosterone
Testosterone has direct vascular actions. It promotes endothelial nitric oxide synthase activity, which supports endothelium-dependent vasodilation. It modulates vascular smooth muscle calcium channel activity, contributing to coronary and peripheral vasodilation. It influences inflammatory tone in the vascular wall, with low testosterone associated with elevated CRP and IL-6 in many cohorts.34,50,56 At the level of the cardiomyocyte, androgen receptors are expressed and physiological androgen support appears to be permissive for normal myocardial energetics.
Testosterone also interacts with the lipoprotein system in ways that are clinically relevant for ASCVD risk. Supraphysiological testosterone, including most anabolic-steroid abuse patterns, lowers HDL and can raise ApoB-containing lipoproteins. Physiological replacement, by contrast, typically produces small reductions in total cholesterol and ApoB and modest reductions in HDL, with neutral or favorable effects on insulin sensitivity and visceral fat.49,50,56 The impact on plaque biology from restoration of normal physiology, as opposed to supraphysiologic exposure, is the variable that matters for prevention.
6.2 Endogenous testosterone and cardiovascular outcomes
Large prospective cohorts have generally found that low endogenous testosterone is associated with increased cardiovascular and all-cause mortality in men. The pooled analysis by Yeap and colleagues, which combined 11 prospective cohorts and over 24,000 men, reported that men in the lowest quintile of total testosterone had a higher risk of cardiovascular events and that very low free testosterone (below approximately 184 pmol/L, or 53 pg/mL) carried the greatest risk.34 A meta-analysis by Corona and colleagues found similar inverse associations between endogenous testosterone and cardiovascular events.35 Mendelian randomization analyses have produced more mixed results but generally do not support a causal harmful effect of physiological testosterone on cardiovascular outcomes, and some analyses suggest a protective association at the lowest end of the distribution.55
SHBG itself appears to be an independent cardiovascular and metabolic biomarker. Low SHBG predicts incident type 2 diabetes,18 and very high SHBG in older men has been associated with mortality in some cohorts, although interpretation is complicated by reverse causation (frailty and undernutrition raise SHBG).19,58
6.3 Testosterone replacement and cardiovascular risk: current evidence after TRAVERSE
In 2013 and 2014, two widely publicized studies (Vigen et al. in the VA system and Finkle et al. in a claims database) reported associations between testosterone replacement therapy (TRT) and cardiovascular events. Both had serious methodologic limitations, including incomplete capture of testosterone monitoring, short follow-up, and selection issues. Subsequent analyses raised methodological concerns about both studies.54,54b Despite this, the FDA in 2015 added a class label warning about possible cardiovascular risk, which reduced prescribing.
Since then, multiple observational studies in better-defined populations have reported neutral or beneficial associations. Sharma and colleagues, in a VA cohort of more than 83,000 men, found that normalization of testosterone with TRT was associated with reduced incidence of myocardial infarction, stroke, and all-cause mortality compared with men whose levels remained low or untreated.37 Cheetham and colleagues reported a 33 percent lower risk of cardiovascular events in TRT-treated men in Kaiser Permanente data.38 Anderson and colleagues, in the Intermountain Healthcare system, similarly reported neutral or favorable cardiovascular outcomes.39
The largest randomized cardiovascular-safety trial to date was TRAVERSE (Lincoff et al., New England Journal of Medicine, 2023), a randomized, placebo-controlled, non-inferiority trial of approximately 5,200 men aged 45 to 80 with hypogonadism and either pre-existing cardiovascular disease or high cardiovascular risk. Over a mean follow-up of 22 months, the primary composite of cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke occurred in 7.0 percent of the testosterone group and 7.3 percent of the placebo group, meeting the prespecified criterion for non-inferiority (hazard ratio 0.96; 95% CI 0.78 to 1.17).36 Atrial fibrillation, acute kidney injury, and pulmonary embolism were modestly more common in the testosterone group, consistent with the known biology of testosterone-induced erythrocytosis; these are the principal residual safety signals requiring monitoring. TRAVERSE was a non-inferiority trial of moderate follow-up duration and does not establish long-term cardiovascular benefit or fully address plaque biology, blood pressure, or broader off-label use.
In 2025, the FDA issued class-wide labeling changes for testosterone products. The updated labels incorporated the TRAVERSE findings and removed boxed-warning language concerning increased risk of major adverse cardiovascular outcomes. The revised labels added or strengthened warnings regarding blood pressure elevation and retained existing limitations of use for age-related hypogonadism in the absence of documented biochemical deficiency. Clinicians should consult the current prescribing information for the specific formulation being used.
6.4 Testosterone and coronary plaque
The Testosterone Trials cardiovascular substudy (Budoff et al., JAMA, 2017) reported that men receiving testosterone gel for one year had a greater increase in non-calcified coronary plaque volume on coronary CT angiography than placebo recipients.48 The trial was not powered for clinical events, the increment in non-calcified plaque was modest, and the longer-term clinical implication remains uncertain. Read alongside TRAVERSE, the most reasonable interpretation is that physiologic restoration has not been shown to increase clinical cardiovascular events at moderate follow-up duration, while plaque-imaging endpoints show signals that warrant continued surveillance.
6.5 Erythrocytosis and thrombotic risk
The most consistently reproducible adverse cardiovascular signal of TRT is erythrocytosis. Testosterone stimulates erythropoiesis through both direct bone marrow effects and increased erythropoietin production, with the magnitude of effect depending on formulation. Injectable esters at peak produce the largest increments; transdermal preparations the smallest. A hematocrit above 54 percent is generally considered an indication to reduce dose, change formulation, or temporarily withhold therapy, and to assess for confounders such as obstructive sleep apnea and dehydration.7,54
6.6 Synthesis for the cardiovascular practitioner
Endogenous testosterone in the low end of the physiologic range is associated with increased cardiovascular risk, almost certainly representing a combination of true biological vulnerability and confounding by adverse metabolic phenotype. Restoration of testosterone to the physiologic range in symptomatic hypogonadal men, monitored appropriately, has not been shown to increase major adverse cardiovascular events at the follow-up durations studied. Erythrocytosis must be monitored. Intensive management of conventional cardiovascular risk factors (ApoB lowering, blood pressure control, glycemic control, smoking cessation, and physical activity) remains the primary cardiovascular intervention. Testosterone replacement is an adjunct that addresses one specific deficiency rather than a cardiovascular therapeutic in its own right.
7. Sexual Desire: The Most Diagnostically Useful Symptom of Testosterone Deficiency
Of all the symptoms attributed to low testosterone, reduced sexual desire is the one most reliably and specifically tied to androgen status. This is not a small detail; it is the most useful diagnostic anchor a clinician has when sorting through the nonspecific complaints that often accompany aging and metabolic disease. It is important to note that the Endocrine Society recommends diagnosing hypogonadism only in men who have symptoms consistent with testosterone deficiency and unequivocally and consistently low testosterone concentrations confirmed on at least two separate morning measurements; symptom status alone is not sufficient.7
7.1 Why reduced sexual desire is the most diagnostically useful symptom
Travison and colleagues, analyzing the Massachusetts Male Aging Study, demonstrated a graded relationship between bioavailable testosterone and libido that holds across the physiological range, while the relationship between testosterone and erectile function is much less consistent.29 EMAS independently identified three sexual symptoms (poor morning erection, low sexual desire, and erectile dysfunction) as the only symptoms that tracked low testosterone with reasonable specificity once age was accounted for.11 Of the three, low desire is the most specific to androgen status; erectile dysfunction is heavily contaminated by vascular, neurogenic, and pharmacologic causes, and morning erections are a sensitive but less specific indicator.
In randomized testosterone-replacement trials, the most consistent and largest effect size is on sexual desire. The Testosterone Trial Sexual Function Substudy (Cunningham et al., 2016) showed that of the three primary sexual outcomes, the gain in desire scores was both the most consistent across men and the most clinically meaningful.30 Meta-analyses of TRT effects on sexual function similarly show that libido is the dominant signal, with smaller and more variable effects on erectile function.32
7.2 What patients describe and what to ask
Men with androgen-deficient libido describe a fundamental drop in the spontaneous interest that previously initiated sexual thought and behavior. They notice they no longer think about sex unprompted, they no longer find ordinary stimuli erotically salient, and they often describe the change as a loss of an internal drive rather than a loss of capability. Men can usually report whether their libido has changed compared to their own past baseline, which is more diagnostically informative than population-referenced thresholds.
Loss of nocturnal and early-morning erections is closely related and often co-reported. The clinical interview should ask about both desire and morning erections explicitly. Asking only about erectile function elicits answers that conflate vascular, neurologic, pharmacologic, and androgen contributions; asking specifically about desire and morning erections separates these.
7.3 What sexual symptoms do not predict
Erectile dysfunction without low desire is rarely resolved by testosterone replacement alone. In men with isolated erectile dysfunction and unequivocally normal bioavailable testosterone, the differential diagnosis is dominated by vascular disease (often the first manifestation of generalized atherosclerosis), neurogenic causes, antihypertensive medications, antidepressants, and psychological factors. Treating these patients with testosterone is rarely effective and exposes them to the side effects of replacement without the corresponding benefit.32,33
8. Other Manifestations of Androgen Deficiency
Beyond sexual symptoms, the syndrome of testosterone deficiency includes physical, metabolic, and psychological features. None is as specific as low libido, but in the right clinical context they corroborate the diagnosis.
8.1 Body composition and metabolism
Hypogonadal men accumulate visceral adipose tissue and lose lean muscle mass, with a corresponding decline in strength.20,21,49,60 This is partly a direct consequence of the loss of androgen signaling at muscle and adipose tissue, and partly a consequence of the metabolic syndrome environment in which obesity-induced hypogonadism develops.59 Long-term replacement studies in obese hypogonadal men have demonstrated meaningful reductions in waist circumference, body weight, and HbA1c; these effects are often more modest than those achieved with intensive lifestyle intervention but appear additive to them.49
8.2 Bone
Testosterone, in part through aromatization to estradiol, supports bone mineral density in men. The Testosterone Trial bone substudy demonstrated significant gains in volumetric bone density and estimated bone strength in older men with low testosterone after one year of replacement.46
8.3 Anemia
Mild anemia is common in untreated hypogonadism. The Testosterone Trial anemia substudy showed correction of anemia in a meaningful proportion of treated men, including some without an identifiable alternative cause.47
8.4 Mood, motivation, and cognition
Low mood, irritability, and reduced sense of vigor are reported by many hypogonadal men and tend to improve modestly with replacement, although these symptoms have many other causes and should not by themselves drive a diagnosis.7,33 Cognitive effects of replacement in older men with mild memory complaints have been small and inconsistent in randomized trials.57
9. Measurement: Methods That Work and Methods That Mislead
9.1 Total testosterone
The Endocrine Society and the CDC Hormone Standardization Program have for over a decade recommended LC-MS/MS as the reference method for total testosterone measurement, particularly at the lower concentrations that matter for diagnosis. Older platform immunoassays are reliable in the mid-to-high adult male range but have substantial error at low concentrations and are not recommended in women.26,27
Pre-analytic variables matter. Total testosterone has a pronounced morning peak and a reduced afternoon trough, with the diurnal amplitude attenuated in older men. Samples should be drawn between 7 and 10 am, fasting, and on at least two separate occasions before a diagnosis of hypogonadism is confirmed. Acute illness, recent strenuous exercise, and recent caloric restriction can transiently lower testosterone and should be considered before interpreting an isolated low value.
9.2 Free testosterone: methods, ranked
Direct analog immunoassays for free testosterone are unreliable and are explicitly not recommended by the major endocrine and andrology societies.26,27 They can produce values that vary several-fold from the true free fraction, with errors largest in the populations where the measurement matters most (men with abnormal SHBG).
Equilibrium dialysis followed by LC-MS/MS is the reference method. The serum sample is dialyzed across a semi-permeable membrane against buffer; only the unbound hormone crosses; the dialysate testosterone is then measured by mass spectrometry. This is the gold standard but is labor-intensive and not widely available.3,28
Calculated free testosterone using the Vermeulen formula has shown excellent agreement with equilibrium dialysis (correlation coefficient r ≈ 0.99 in well-conducted comparisons), provided that total testosterone and SHBG are measured by accurate platforms.3,28 The calculation is the practical method most clinicians should use; the more recent Goldman et al. multistep allosteric model improves accuracy at the extremes of SHBG but is not yet routinely available.2
Table 3. Methods for measuring or estimating free testosterone
| Method | Accuracy | Practical use | Limitation |
| Direct analog immunoassay | Poor | Not recommended | Substantially inaccurate at low and high SHBG; errors largest in the populations where measurement matters most |
| Equilibrium dialysis + LC-MS/MS | Reference standard | Research and diagnostically difficult cases | Labor-intensive, slow, expensive, and not widely available in routine clinical laboratories |
| Vermeulen calculated free T | High (r ≈ 0.99 vs equilibrium dialysis) | Routine clinical use | Dependent on quality of total testosterone and SHBG inputs; less accurate at extremes of SHBG |
| Goldman multistep allosteric model | Highest at extremes of SHBG | Selected difficult cases | Not yet broadly implemented in routine clinical laboratory systems |
10. Restoring Bioavailable Testosterone: Pharmacology
10.1 Therapeutic targets
The aim of replacement is to restore symptoms (with sexual desire as the primary endpoint) while keeping biochemistry within the physiologic range for healthy young men. The Endocrine Society guideline targets a mid-normal total testosterone, typically 400 to 700 ng/dL (14 to 24 nmol/L), with corresponding free testosterone in the mid-normal range.7 In men with elevated SHBG, total testosterone targets must be interpreted with care; bioavailable testosterone, not total, should drive titration in this group.
10.2 Formulations
Topical gels and solutions deliver steady serum levels approximating the physiologic diurnal rhythm. They are first-line in many guidelines for that reason, with the trade-offs of daily application, modest skin reactions, and the small but real risk of interpersonal transfer.7
Intramuscular esters (testosterone cypionate or enanthate) are inexpensive and effective. The classic regimen of 100 to 200 mg every 1 to 2 weeks produces large peaks and troughs; many practitioners now favor smaller, more frequent injections (for instance, 50 to 80 mg twice weekly, often subcutaneously) to flatten the pharmacokinetic profile and reduce hematocrit excursions.7,8
Long-acting testosterone undecanoate, given by deep intramuscular injection every 10 to 14 weeks after a loading phase, produces stable levels and is widely used outside the United States. The US formulation requires in-office observation due to the historically rare risk of pulmonary oil microembolism.51
Subcutaneous testosterone pellets, implanted every 3 to 6 months, provide stable levels for men who prefer infrequent dosing.8
Oral testosterone undecanoate (absorbed via the lymphatic route in current US formulations) and intranasal testosterone gel are additional options, each with specific monitoring considerations.
10.3 Dosing in high versus low SHBG
In men with high SHBG (often older, lean, with subclinical hyperthyroidism, or on anticonvulsants) higher doses may be needed to achieve a satisfactory bioavailable level, and the total testosterone may need to run in the high-normal range. In men with low SHBG (typically obese or insulin-resistant) standard doses may produce excessive free testosterone if titrated to total testosterone alone, leading to acne, irritability, and accelerated erythrocytosis. Calculated bioavailable testosterone should drive titration in both populations.
11. Human Chorionic Gonadotropin: Mechanism and Clinical Use
Human chorionic gonadotropin (hCG) acts as an analog of luteinizing hormone, binding the LH receptor on Leydig cells and stimulating intratesticular testosterone production directly. Because exogenous testosterone suppresses pituitary LH and FSH and shuts down endogenous Leydig cell stimulation (with the consequence that intratesticular testosterone falls and spermatogenesis is suppressed), hCG plays two important clinical roles: as monotherapy in selected men, and as an adjunct to testosterone replacement when fertility preservation matters.
11.1 The intratesticular testosterone problem
Spermatogenesis requires intratesticular testosterone concentrations roughly 50 to 100 times higher than circulating testosterone. Exogenous testosterone, by suppressing LH, removes the stimulus for intratesticular testosterone production. Coviello and colleagues demonstrated that low-dose hCG (250 IU subcutaneously every other day) administered concomitantly with exogenous testosterone preserves intratesticular testosterone within the normal range, while testosterone alone reduces it by more than 90 percent.41
Hsieh and colleagues subsequently showed that men receiving testosterone replacement with concomitant low-dose hCG (typically 500 IU three times weekly) maintained semen parameters across treatment, while those without hCG developed expected oligospermia or azoospermia.42 Ramasamy and others have summarized the practical implementation of fertility-preserving combination therapy.43
11.2 hCG monotherapy
In younger hypogonadal men with secondary or mixed hypogonadism who wish to maintain fertility, hCG monotherapy is a reasonable first choice. Typical dosing begins at 1,000 to 1,500 IU subcutaneously two to three times per week, titrated against total testosterone, free testosterone, and estradiol response. Many men achieve mid-normal total testosterone on monotherapy. Estradiol may rise more than with testosterone replacement (because testicular aromatization is preserved), and selective use of an aromatase inhibitor is occasionally warranted; this should not be reflexive.41,43,44
Use of hCG in this setting is generally off-label in most jurisdictions and should be individualized, ideally with reproductive-urology involvement when fertility is an active goal. Evidence supporting hCG protocols derives primarily from mechanistic studies and small observational or prospective trials rather than large randomized controlled trials.
11.3 hCG adjunctive to testosterone replacement
In men on testosterone replacement who wish to preserve fertility, or who experience uncomfortable testicular atrophy, low-dose hCG (typically 500 IU two or three times weekly) is added to the testosterone regimen. This dose preserves intratesticular testosterone and spermatogenesis without producing excessive total testosterone or estradiol elevations.41,42,43 As with monotherapy, this combination use is generally off-label and the supporting evidence is largely observational.
11.4 Restart protocols after testosterone withdrawal
Men who have used exogenous testosterone (whether prescribed or as part of anabolic-androgen use) and who wish to recover endogenous function and fertility are candidates for restart protocols. The components typically include hCG to drive Leydig cell recovery, a selective estrogen receptor modulator (clomiphene or tamoxifen) to disinhibit pituitary gonadotropin secretion, and in some cases recombinant FSH if spermatogenesis fails to recover. Wenker and colleagues reported encouraging recovery rates with such combinations in men attempting to resume fertility after exogenous androgen exposure.44,45
11.5 Practical considerations
hCG is administered by subcutaneous injection. Side effects are usually mild and include local injection-site reactions, mild gynecomastia (when estradiol rises), and acne. Cost has historically been variable, and compounded formulations are available in some jurisdictions, where quality control is a legitimate clinical concern.
12. Monitoring of Therapy
Initiation and titration require systematic surveillance. The following framework is consistent with the Endocrine Society and AUA guidelines, modified by the cardiovascular evidence reviewed above.7,8
Baseline. Total testosterone (LC-MS/MS or validated platform assay), SHBG, calculated free or bioavailable testosterone (Vermeulen), albumin if the calculation is to be performed, LH and FSH (to distinguish primary from secondary hypogonadism), prolactin (in selected cases), TSH, complete blood count (with attention to hematocrit), comprehensive metabolic panel, fasting lipid panel including ApoB where available, PSA in men over 40, and digital rectal examination per AUA guidance.
Three months. Symptom review (with sexual desire as the primary endpoint), repeat total testosterone (timed correctly for the formulation: trough for intramuscular esters, 2 to 8 hours post-application for gels), CBC, and PSA where appropriate. Hematocrit above 54 percent triggers dose reduction or formulation change.
Six and twelve months. Same panel, plus reassessment of body composition and waist circumference, lipid response, and glycemic indices in men with metabolic disease.
Annual thereafter. Symptom review, total and free testosterone, hematocrit, PSA in men over 40, and comprehensive cardiovascular risk reassessment. PSA increases of more than 1.4 ng/mL within 12 months, or a confirmed PSA above 4.0 ng/mL, warrant urological evaluation.7,8,53
13. Safety: What to Watch and What Has Been Resolved
13.1 Prostate
Modern data do not support the older notion that physiological testosterone replacement causes prostate cancer in men with normal baseline prostate health. The saturation model proposed by Morgentaler and colleagues, in which androgen receptor signaling is saturated within the physiological range, fits the available data better than the linear stimulation model implicit in older recommendations.52,53 This does not mean replacement is safe in untreated prostate cancer; it means that uncomplicated benign prostatic hyperplasia and a stable PSA do not contraindicate replacement when symptomatic hypogonadism is documented.
13.2 Erythrocytosis
Discussed in Section 6.5. Erythrocytosis is the most common dose-limiting laboratory abnormality of TRT. Coexisting obstructive sleep apnea should be sought in men with persistent erythrocytosis on appropriate doses.
13.3 Fertility
Exogenous testosterone reliably suppresses spermatogenesis. Men of reproductive age must be counseled in advance, and hCG-based combination therapy should be offered to those who wish to preserve fertility.41,42,43,45
13.4 Cardiovascular
Discussed in detail in Section 6. The TRAVERSE trial provides Level 1 evidence that physiological replacement in symptomatic hypogonadal men, including those with elevated cardiovascular risk, has not been shown to increase major adverse cardiovascular events at the follow-up durations studied.36 The 2025 FDA labeling update reflects this evidence. Erythrocytosis, atrial fibrillation, blood pressure, and venous thromboembolism remain monitorable signals.
13.5 Other considerations
Acne, mild gynecomastia (when estradiol rises), and edema are dose-related and usually manageable. Sleep apnea may worsen at high doses. Mood changes (typically improvement, occasionally irritability at supraphysiologic levels) should be tracked alongside laboratory parameters.
14. Conclusions
The diagnosis of testosterone deficiency in men is fundamentally a problem of identifying tissue-level androgen exposure, not of measuring a number on a laboratory report. Total testosterone is a screening tool, useful in young men with healthy SHBG biology, frequently misleading in older men or in any patient with metabolic disease. Bioavailable testosterone, calculated reliably from a high-quality total testosterone measurement and a measured SHBG, is the variable that matters.
SHBG itself deserves more clinical attention than it usually receives. It rises with age, with hyperthyroidism, with estrogen exposure, with caloric restriction, and with hepatic dysfunction. It falls with hyperinsulinemia, obesity, and anabolic steroid use. The principal non-pharmacologic levers for lowering clinically elevated SHBG are improving insulin sensitivity, addressing thyroid abnormalities, transitioning oral estrogens to transdermal where indicated, ensuring adequate caloric and protein intake, and treating any underlying hepatic process.
Clinically, sexual desire is the single most diagnostically useful symptom of androgen deficiency, provided it is accompanied by biochemical confirmation of consistently low testosterone on at least two morning measurements. A man with low desire and morning-erection loss in the context of confirmed low bioavailable testosterone is the patient most likely to benefit from replacement; a man with isolated erectile dysfunction and normal bioavailable testosterone is the patient least likely to benefit and most likely to need cardiovascular evaluation instead.
The cardiovascular dimension has come into focus over the last decade. Low endogenous testosterone correlates with increased cardiovascular and total mortality. Restoration to the physiologic range, monitored for erythrocytosis and blood pressure, has not been shown to increase major adverse cardiovascular events in the largest available randomized trial (TRAVERSE), and may in selected populations be associated with risk reduction in observational data. The 2025 FDA labeling update reflects this evolving evidence. Replacement therapy is not a substitute for ApoB lowering and lifestyle intervention; it is an adjunct that addresses one specific contributor to cardiovascular and metabolic risk in men who have a true deficiency.
Therapeutic restoration is achieved with testosterone preparations chosen to match the patient’s lifestyle and pharmacokinetic needs. In men of reproductive age, in men who object to testicular atrophy, or in men whose hypogonadism is secondary and who wish to maintain fertility, hCG (alone or in combination with testosterone) is the more appropriate strategy. The clinician’s task is to match the physiology to the patient and to monitor diligently.
Acknowledgments
The author acknowledges the use of an AI language model (Claude, Anthropic) in the drafting, structural organization, and editorial revision of this manuscript. All scientific content, clinical interpretations, literature citations, and the underlying research synthesis represent the work and judgment of the author. All cited sources were independently identified and verified by the author. The AI was used as a writing and editing tool; it did not contribute to the intellectual or scientific substance of the review. This disclosure is made in accordance with emerging editorial standards for AI-assisted manuscript preparation.
References
- Hammond GL. Diverse roles for sex hormone-binding globulin in reproduction. Biol Reprod. 2011;85(3):431-441. doi:10.1095/biolreprod.111.092593
- Goldman AL, Bhasin S, Wu FCW, Krishna M, Matsumoto AM, Jasuja R. A reappraisal of testosterone’s binding in circulation: physiological and clinical implications. Endocr Rev. 2017;38(4):302-324. doi:10.1210/er.2017-00025
- Vermeulen A, Verdonck L, Kaufman JM. A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab. 1999;84(10):3666-3672. doi:10.1210/jcem.84.10.6079
- Mendel CM. The free hormone hypothesis: a physiologically based mathematical model. Endocr Rev. 1989;10(3):232-274. doi:10.1210/edrv-10-3-232
- Selva DM, Hogeveen KN, Innis SM, Hammond GL. Monosaccharide-induced lipogenesis regulates the human hepatic sex hormone-binding globulin gene. J Clin Invest. 2007;117(12):3979-3987. doi:10.1172/JCI32249
- Pugeat M, Nader N, Hogeveen K, Raverot G, Déchaud H, Grenot C. Sex hormone-binding globulin gene expression in the liver: drugs and the metabolic syndrome. Mol Cell Endocrinol. 2010;316(1):53-59. doi:10.1016/j.mce.2009.09.020
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103(5):1715-1744. doi:10.1210/jc.2018-00229
- Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200(2):423-432. doi:10.1016/j.juro.2018.03.115
- Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR. Longitudinal effects of aging on serum total and free testosterone levels in healthy men: Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab. 2001;86(2):724-731. doi:10.1210/jcem.86.2.7219
- Feldman HA, Longcope C, Derby CA, et al. Age trends in the level of serum testosterone and other hormones in middle-aged men: longitudinal results from the Massachusetts Male Aging Study. J Clin Endocrinol Metab. 2002;87(2):589-598. doi:10.1210/jcem.87.2.8201
- Wu FCW, Tajar A, Beynon JM, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363(2):123-135. doi:10.1056/NEJMoa0911101
- Travison TG, Vesper HW, Orwoll E, et al. Harmonized reference ranges for circulating testosterone levels in men of four cohort studies in the United States and Europe. J Clin Endocrinol Metab. 2017;102(4):1161-1173. doi:10.1210/jc.2016-2935
- Kaufman JM, Vermeulen A. The decline of androgen levels in elderly men and its clinical and therapeutic implications. Endocr Rev. 2005;26(6):833-876. doi:10.1210/er.2004-0013
- Veldhuis JD, Keenan DM, Liu PY, Iranmanesh A, Takahashi PY, Nehra AX. The aging male hypothalamic-pituitary-gonadal axis: pulsatility and feedback. Mol Cell Endocrinol. 2009;299(1):14-22. doi:10.1016/j.mce.2008.09.005
- Camacho EM, Huhtaniemi IT, O’Neill TW, et al. Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: longitudinal results from the European Male Ageing Study. Eur J Endocrinol. 2013;168(3):445-455. doi:10.1530/EJE-12-0890
- Travison TG, Araujo AB, O’Donnell AB, Kupelian V, McKinlay JB. A population-level decline in serum testosterone levels in American men. J Clin Endocrinol Metab. 2007;92(1):196-202. doi:10.1210/jc.2006-1375
- Selva DM, Hammond GL. Thyroid hormones act indirectly to increase sex hormone-binding globulin production by liver via hepatocyte nuclear factor-4alpha. J Mol Endocrinol. 2009;43(1):19-27. doi:10.1677/JME-09-0025
- Ding EL, Song Y, Manson JE, et al. Sex hormone-binding globulin and risk of type 2 diabetes in women and men. N Engl J Med. 2009;361(12):1152-1163. doi:10.1056/NEJMoa0804381
- Brand JS, van der Tweel I, Grobbee DE, Emmelot-Vonk MH, van der Schouw YT. Testosterone, sex hormone-binding globulin and the metabolic syndrome: a systematic review and meta-analysis of observational studies. Int J Epidemiol. 2011;40(1):189-207. doi:10.1093/ije/dyq158
- Kelly DM, Jones TH. Testosterone and obesity. Obes Rev. 2015;16(7):581-606. doi:10.1111/obr.12282
- Grossmann M. Hypogonadism and male obesity: focus on unresolved questions. Clin Endocrinol (Oxf). 2018;89(1):11-21. doi:10.1111/cen.13723
- Saboor Aftab SA, Kumar S, Barber TM. The role of obesity and type 2 diabetes mellitus in the development of male obesity-associated secondary hypogonadism. Clin Endocrinol (Oxf). 2013;78(3):330-337. doi:10.1111/cen.12092
- Dhindsa S, Ghanim H, Batra M, et al. Hypogonadotropic hypogonadism in men with diabesity. Diabetes Care. 2018;41(7):1516-1525. doi:10.2337/dc17-2510
- Sinclair M, Grossmann M, Gow PJ, Angus PW. Testosterone in men with advanced liver disease: abnormalities and implications. J Gastroenterol Hepatol. 2015;30(2):244-251. doi:10.1111/jgh.12695
- Schiavi A, Carugno M, Tozzi V, et al. Endocrine dysfunction in nephrotic syndrome: a comprehensive review. Biomedicines. 2024;12(8):1860. doi:10.3390/biomedicines12081860
- Rosner W, Auchus RJ, Azziz R, Sluss PM, Raff H. Position statement: utility, limitations, and pitfalls in measuring testosterone: an Endocrine Society position statement. J Clin Endocrinol Metab. 2007;92(2):405-413. doi:10.1210/jc.2006-1864
- Rosner W, Vesper H. Toward excellence in testosterone testing: a consensus statement. J Clin Endocrinol Metab. 2010;95(10):4542-4548. doi:10.1210/jc.2010-1314
- Fiers T, Wu F, Moghetti P, Vanderschueren D, Lapauw B, Kaufman JM. Reassessing free-testosterone calculation by liquid chromatography-tandem mass spectrometry direct equilibrium dialysis. J Clin Endocrinol Metab. 2018;103(6):2167-2174. doi:10.1210/jc.2017-02360
- Travison TG, Morley JE, Araujo AB, O’Donnell AB, McKinlay JB. The relationship between libido and testosterone levels in aging men. J Clin Endocrinol Metab. 2006;91(7):2509-2513. doi:10.1210/jc.2005-2508
- Cunningham GR, Stephens-Shields AJ, Rosen RC, et al. Testosterone treatment and sexual function in older men with low testosterone levels. J Clin Endocrinol Metab. 2016;101(8):3096-3104. doi:10.1210/jc.2016-1645
- Snyder PJ, Bhasin S, Cunningham GR, et al. Effects of testosterone treatment in older men. N Engl J Med. 2016;374(7):611-624. doi:10.1056/NEJMoa1506119
- Corona G, Isidori AM, Buvat J, et al. Testosterone supplementation and sexual function: a meta-analysis study. J Sex Med. 2014;11(6):1577-1592. doi:10.1111/jsm.12536
- Buvat J, Maggi M, Gooren L, et al. Endocrine aspects of male sexual dysfunctions. J Sex Med. 2010;7(4 Pt 2):1627-1656. doi:10.1111/j.1743-6109.2010.01780.x
- Yeap BB, Marriott RJ, Antonio L, et al. Associations of serum testosterone and sex hormone-binding globulin with incident cardiovascular events in middle-aged to older men. Ann Intern Med. 2022;175(2):159-170. doi:10.7326/M21-0551
- Corona G, Rastrelli G, Di Pasquale G, Sforza A, Mannucci E, Maggi M. Endogenous testosterone levels and cardiovascular risk: meta-analysis of observational studies. J Sex Med. 2018;15(9):1260-1271. doi:10.1016/j.jsxm.2018.06.012
- Lincoff AM, Bhasin S, Flevaris P, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389(2):107-117. doi:10.1056/NEJMoa2215025
- Sharma R, Oni OA, Gupta K, et al. Normalization of testosterone level is associated with reduced incidence of myocardial infarction and mortality in men. Eur Heart J. 2015;36(40):2706-2715. doi:10.1093/eurheartj/ehv346
- Cheetham TC, An J, Jacobsen SJ, et al. Association of testosterone replacement with cardiovascular outcomes among men with androgen deficiency. JAMA Intern Med. 2017;177(4):491-499. doi:10.1001/jamainternmed.2016.9546
- Anderson JL, May HT, Lappe DL, et al. Impact of testosterone replacement therapy on myocardial infarction, stroke, and death in men with low testosterone concentrations in an integrated health care system. Am J Cardiol. 2016;117(5):794-799. doi:10.1016/j.amjcard.2015.11.063
- Mohammed M, Al-Habori M, Abdullateef A, Saif-Ali R. Impact of metabolic syndrome factors on testosterone and SHBG in type 2 diabetes mellitus and metabolic syndrome. J Diabetes Res. 2018;2018:4926789. doi:10.1155/2018/4926789
- Coviello AD, Matsumoto AM, Bremner WJ, et al. Low-dose human chorionic gonadotropin maintains intratesticular testosterone in normal men with testosterone-induced gonadotropin suppression. J Clin Endocrinol Metab. 2005;90(5):2595-2602. doi:10.1210/jc.2004-0802
- Hsieh TC, Pastuszak AW, Hwang K, Lipshultz LI. Concomitant intramuscular human chorionic gonadotropin preserves spermatogenesis in men undergoing testosterone replacement therapy. J Urol. 2013;189(2):647-650. doi:10.1016/j.juro.2012.09.043
- Ramasamy R, Armstrong JM, Lipshultz LI. Preserving fertility in the hypogonadal patient: an update. Asian J Androl. 2015;17(2):197-200. doi:10.4103/1008-682X.142772
- Wenker EP, Dupree JM, Langille GM, et al. The use of HCG-based combination therapy for recovery of spermatogenesis after testosterone use. J Sex Med. 2015;12(6):1334-1337. doi:10.1111/jsm.12890
- Crosnoe LE, Grober E, Ohl D, Kim ED. Exogenous testosterone: a preventable cause of male infertility. Transl Androl Urol. 2013;2(2):106-113. doi:10.3978/j.issn.2223-4683.2013.06.01
- Snyder PJ, Kopperdahl DL, Stephens-Shields AJ, et al. Effect of testosterone treatment on volumetric bone density and strength in older men with low testosterone: a controlled clinical trial. JAMA Intern Med. 2017;177(4):471-479. doi:10.1001/jamainternmed.2016.9539
- Roy CN, Snyder PJ, Stephens-Shields AJ, et al. Association of testosterone levels with anemia in older men: a controlled clinical trial. JAMA Intern Med. 2017;177(4):480-490. doi:10.1001/jamainternmed.2016.9540
- Budoff MJ, Ellenberg SS, Lewis CE, et al. Testosterone treatment and coronary artery plaque volume in older men with low testosterone. JAMA. 2017;317(7):708-716. doi:10.1001/jama.2016.21043
- Saad F, Yassin A, Doros G, Haider A. Effects of long-term treatment with testosterone on weight and waist size in 411 hypogonadal men with obesity classes I-III: observational data from two registry studies. Int J Obes (Lond). 2016;40(1):162-170. doi:10.1038/ijo.2015.139
- Traish AM. Benefits and health implications of testosterone therapy in men with testosterone deficiency. Sex Med Rev. 2018;6(1):86-105. doi:10.1016/j.sxmr.2017.10.001
- Hackett G, Cole N, Bhartia M, Kennedy D, Raju J, Wilkinson P. Testosterone replacement therapy with long-acting testosterone undecanoate improves sexual function and quality-of-life parameters versus placebo in a population of men with type 2 diabetes. J Sex Med. 2013;10(6):1612-1627. doi:10.1111/jsm.12146
- Morgentaler A, Zitzmann M, Traish AM, et al. Fundamental concepts regarding testosterone deficiency and treatment: international expert consensus resolutions. Mayo Clin Proc. 2016;91(7):881-896. doi:10.1016/j.mayocp.2016.04.007
- Khera M, Crawford D, Morales A, Salonia A, Morgentaler A. A new era of testosterone and prostate cancer: from physiology to clinical implications. Eur Urol. 2014;65(1):115-123. doi:10.1016/j.eururo.2013.08.015
- Glueck CJ, Wang P. Testosterone therapy, thrombosis, thrombophilia, cardiovascular events. Metabolism. 2014;63(8):989-994. doi:10.1016/j.metabol.2014.05.005
54b. Baillargeon J, Urban RJ, Kuo YF, et al. Risk of myocardial infarction in older men receiving testosterone therapy. Ann Pharmacother. 2014;48(9):1138-1144. doi:10.1177/1060028014539918
- Mohammadi-Shemirani P, Chong M, Pigeyre M, et al. Effects of lifelong testosterone exposure on health and disease using Mendelian randomization. eLife. 2020;9:e58914. doi:10.7554/eLife.58914
- Mohler ER 3rd, Ellenberg SS, Lewis CE, et al. The effect of testosterone on cardiovascular biomarkers in the testosterone trials. J Clin Endocrinol Metab. 2018;103(2):681-688. doi:10.1210/jc.2017-02243
- Resnick SM, Matsumoto AM, Stephens-Shields AJ, et al. Testosterone treatment and cognitive function in older men with low testosterone and age-associated memory impairment. JAMA. 2017;317(7):717-727. doi:10.1001/jama.2016.20828
- Yeap BB, Knuiman MW, Divitini ML, et al. Differential associations of testosterone, dihydrotestosterone and oestradiol with physical, metabolic and health-related factors in community-dwelling men aged 17-97 years from the Busselton Health Survey. Clin Endocrinol (Oxf). 2014;81(1):100-108. doi:10.1111/cen.12407
- Kupelian V, Page ST, Araujo AB, Travison TG, Bremner WJ, McKinlay JB. Low sex hormone-binding globulin, total testosterone, and symptomatic androgen deficiency are associated with development of the metabolic syndrome in nonobese men. J Clin Endocrinol Metab. 2006;91(3):843-850. doi:10.1210/jc.2005-1326
- Allan CA, Strauss BJ, Burger HG, Forbes EA, McLachlan RI. Testosterone therapy prevents gain in visceral adipose tissue and loss of skeletal muscle in nonobese aging men. J Clin Endocrinol Metab. 2008;93(1):139-146. doi:10.1210/jc.2007-1291