The Big Pipe: The Guide to the Aorta and Staying Active
-
The Body’s Super-Highway
Imagine your body is a giant, bustling city. For the city to work, it needs one massive main water pipe to carry energy and supplies to every single house. In your body, that “water main” is a giant tube called the aorta. Doctors call it the “Great Vessel” because it is the biggest and most important power line in your system. It carries all the fresh, oxygen-rich blood from your heart down to your toes, up to your brain, and everywhere in between.
Because this pipe is the primary highway for your blood, it needs to be incredibly strong to handle the “traffic” pumping through it every second. For people who love to play sports, run, or lift heavy things, the aorta has to be extra tough to handle the extra flow. However, sometimes this big pipe can develop a weak spot. This “silent” problem is called an aneurysm, and understanding how it works is the secret to staying safe while staying active.
-
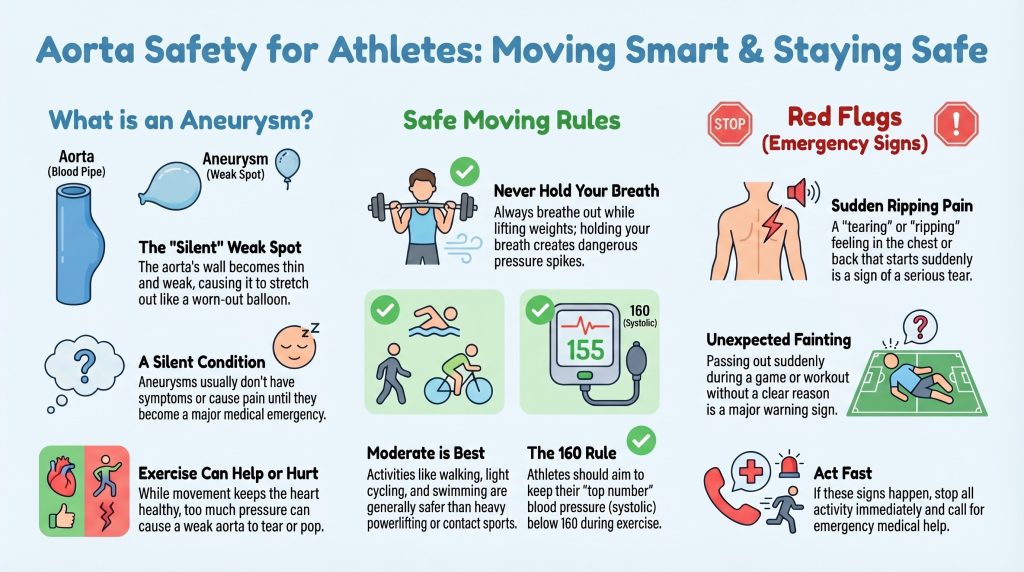
What is an Aneurysm? (The Balloon Effect)
An aneurysm happens when a section of the aorta gets a “bulge,” making it look like a balloon starting to inflate. Doctors officially call it an aneurysm when the pipe gets 50% wider than its normal size. To understand why this is a problem, we look at Laplace’s Law.
Think of a balloon: as you blow it up, the rubber gets wider, but it also gets much thinner. The more it stretches, the easier it is to pop. In the aorta, as the “tube” gets wider, the walls get thinner and weaker. Because your heart is a powerful pump, that thin wall has to work much harder to hold back the high-pressure blood.
Why the Bulge is Dangerous
- Thinned Walls: The “building blocks” of the pipe (like elastic fibers) are stretched out or broken down.
- High Pressure: Every heartbeat pushes against the weak spot like a finger poking a thin, over-inflated balloon.
- The Silent Challenge: The biggest problem is that these bulges usually don’t hurt! A person can have a weak spot for years without knowing it, which is why doctors have to be “Detectives” to find them before they cause trouble.
-
A Tale of Two Tunnels: Tummy vs. Chest
Not all weak spots are the same. Doctors treat the “tummy” area and the “chest” area as two different worlds because they speak different “biological languages.”
| Feature | Abdominal Aortic Aneurysm (AAA) | Thoracic Aortic Aneurysm (TAA) |
| Location | The Tummy | The Chest |
| Main Cause | Pipe Clogging (Atherosclerosis) | Weak Middle Layer (Genetics) |
| The “Gunk” | Clogged up with “rust” (plaque) and swelling. | The building materials wear out early. |
| The Science | Swelling caused by “Molecular Scissors” (MMPs) that cut up the pipe’s lining. | Often linked to “instruction manual” errors in your DNA (like Marfan Syndrome). |
Because the causes are so different, a doctor’s plan depends entirely on where the bulge is. For athletes, these spots have to handle a lot of extra “traffic” whenever the game starts.
-
Sports and the “Pressure Test”
When you exercise, your heart pumps harder. But different types of play affect your “pipes” in different ways.
- Running and Cycling (The Traffic Load): This sends a “high volume” of blood through the pipes for a long time. It’s like a highway having five times more cars than usual for an hour.
- Heavy Lifting (The Rebound Spike): When someone lifts something very heavy and holds their breath (the Valsalva Paradox), the pressure inside the chest goes way up. Surprisingly, the most dangerous moment isn’t the lift itself—it’s the “rebound” the second you let your breath out. This creates a “backward wave” of pressure that can snap against a weak aorta wall.
Key Finding: Wall Stress Data Research shows that during moderate exercise, the stress on the aorta wall is about 257–323 kPa. (Think of kPa like the air pressure in a bike tire—it’s just a way to measure how hard the blood is pushing). This is very safe, as the “breaking point” for a weak aorta is much higher, between 850–1,200 kPa. Moving is usually great for you if you don’t overdo the pressure!
-
Is it a “Strong” Pipe or a “Sick” Pipe?
Doctors have a tricky job because elite athletes naturally have larger hearts and pipes—it’s how their bodies adapt to being strong! On average, an elite athlete’s aorta is 1.69 mm larger than a non-athlete’s.
To figure out if a pipe is “big because it’s strong” or “big because it’s sick,” doctors use Indexing. Think of this like Sizing for Clothes:
- A “Large” shirt is perfectly normal for a grown-up basketball player, but it would be a “Red Flag” for a toddler.
- A tall athlete should have a bigger aorta than a smaller person.
Doctors use a “math grade” called a Z-score to see if the pipe fits the person’s body size. If the measurement is too high (like over 42 mm for a young man), the “Detective” starts looking closer to make sure the athlete stays safe on the field.
-
The Safety Rulebook: How to Keep Playing
In the old days, doctors might have told someone with a weak aorta to stop all sports. Today, they use “precision” to help athletes keep moving safely.
Safety Rules for Aorta Health
- No Holding Your Breath: Always keep breathing while lifting or playing to avoid that “backward wave” pressure spike.
- Target “Moderate” Effort: Use the Borg Scale (a 1-10 scale of how hard you feel you’re working). Aim for a level where you are working hard but not gasping for air.
- Blood Pressure Ceilings: Keep your “blood push” under control (doctors usually want it under 160/120 during activity).
- The Pilot & Co-Pilot Rule: This is called “Shared Decision-Making.” You are the Pilot of your body, and the doctor is your Co-Pilot. You work together as a team to navigate a safe flight.
-
The Emergency Alarm: Knowing When to Stop
Even with the best rules, you must know when there is a “Level 10” emergency. If a weak aorta actually starts to tear, it is called a dissection.
How to tell the difference:
- Tearing vs. Soreness: A pulled muscle usually feels stiff or builds up slowly. An aortic tear is a “tearing or ripping” pain that is at its absolute worst (Level 10) the very first second it starts.
- The Pulse Clue: You might notice a strong pulse in one arm but a very weak or missing one in the other.
- Other Alarms: Fainting for no reason or a doctor hearing a new “whooshing” sound in the heart (a murmur).
If the “Emergency Alarm” goes off, the rule is simple: Stop everything immediately and get to a hospital right away.
-
Conclusion: Smart Moving, Strong Heart
We used to think that a weak aorta meant the end of sports. Now we know better. The aorta is a complex machine that needs the right amount of “traffic” to stay healthy.
By moving away from just saying “no” and moving toward “playing smart,” we can keep athletes on the field. Your knowledge—from understanding your DNA to keeping an eye on your blood pressure—is the best tool you have to keep your “Big Pipe” strong and your heart moving for a lifetime.
DEEP DIVE
CLINICAL ANALYSIS
When the Heart’s Great Vessel Fails: Aortic Aneurysm and the Athlete
A comprehensive synthesis of pathophysiology, hemodynamic biomechanics, and evidence-based guidelines for athletic participation in patients with aortic disease.
| Aortic aneurysms represent one of cardiovascular medicine’s most formidable diagnostic and management challenges: a condition that kills silently, strikes catastrophically, and demands nuanced, individualized clinical judgment. For the competitive athlete harboring an underlying aortopathy—often undiagnosed—the intersection of molecular pathology and intense hemodynamic stress creates a uniquely high-stakes clinical dilemma. This review examines the dual biological paradigms driving abdominal and thoracic aortic disease, quantifies the biomechanical demands imposed by distinct athletic modalities, and synthesizes the current evidence base to define the boundary between life-enhancing physical activity and lethal mechanical risk. |
SECTION I
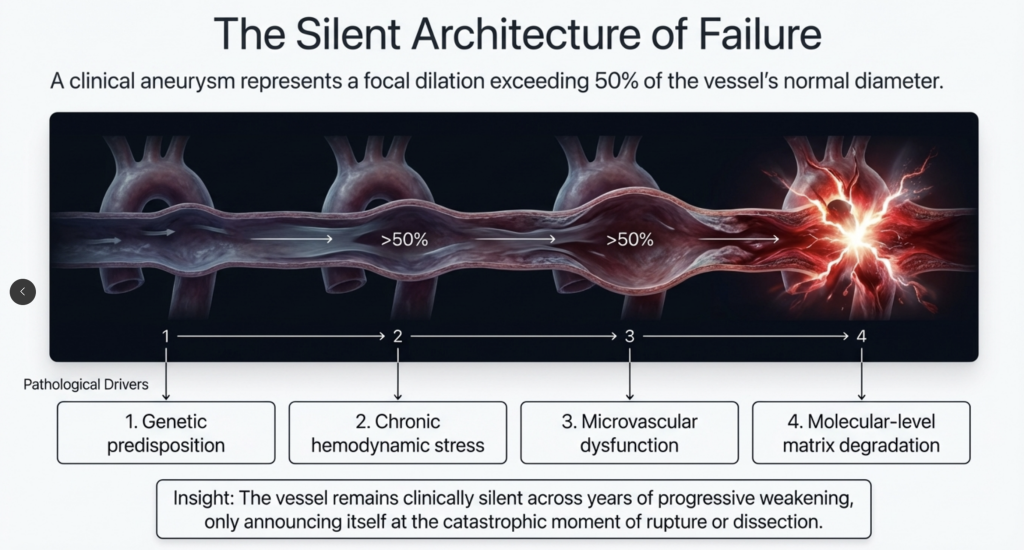
The Silent Architecture of Failure
A clinically defined aortic aneurysm—a focal dilation exceeding 50% of the vessel’s normal diameter—is among the most deceptive entities in cardiovascular disease. Its pathogenesis is multifactorial, driven by the convergence of genetic predisposition, chronic hemodynamic stress, microvascular dysfunction, and molecular-level matrix degradation. The vessel remains clinically silent across years of progressive weakening, only announcing itself at the catastrophic moment of rupture or dissection.[1]
The clinical imperative to understand aortic disease in the context of athletics has never been sharper. International cardiology societies universally endorse regular physical activity as a cornerstone intervention for the systemic cardiovascular risk factors—hypertension, dyslipidemia, atherosclerosis—that simultaneously drive aortic wall failure.[2] Yet the same intense exertion that protects the heart can, in a compromised aorta, translate directly into catastrophic mechanical loading. For the sports cardiologist, resolving this tension is not merely an academic exercise; it is a matter of life and death.
SECTION II
Two Diseases, Two Biological Languages
Accurate risk stratification begins with a foundational distinction that the clinical literature has only recently made consistently clear: abdominal aortic aneurysms (AAA) and thoracic aortic aneurysms (TAA) are not the same disease wearing different anatomical addresses. Their initiating pathophysiological cascades, molecular signatures, and clinical behaviors are markedly distinct, demanding entirely separate conceptual frameworks.[1]
Abdominal Aortic Aneurysms: An Inflammatory–Proteolytic Disease
AAA is, at its core, a disease of chronic systemic inflammation amplified by atherosclerotic burden. Extensive population analyses—including cohorts of more than 6,400 participants aged 25 to 74 years—have established a significant statistical association between carotid artery plaque burden and AAA prevalence, cementing atherosclerosis as a systemic harbinger of abdominal wall failure.[3] The shared epidemiological profile with coronary and peripheral arterial disease is not coincidental; it is mechanistic.[4]
The molecular engine of AAA destruction is a profoundly imbalanced proteolytic cascade. The structural integrity of the healthy aortic wall depends on a tightly woven scaffold of elastin and collagen. In active aneurysmal disease, this matrix is irreversibly dismantled by an unchecked surge of Matrix Metalloproteinases—specifically MMP-2 and MMP-9—operating in the near-absence of their endogenous inhibitors, TIMP-1 and TIMP-2.[1]
This proteolytic environment is created and sustained by deep leukocytic infiltration. Activated macrophages, T lymphocytes, and B cells invade the aortic media and adventitia, continuously secreting pro-inflammatory cytokines including IL-6, IL-1β, and TNF-α.[1] A particularly well-characterized molecular feature of AAA pathogenesis is the activation of the NLRP3 inflammasome, which perpetuates the localized inflammatory storm within the abdominal vessel wall.[1] Concurrently, medial smooth muscle cells abandon their contractile phenotype, halting matrix production and instead contributing to further MMP secretion—essentially cannibalizing their own structural framework. Chronic activation of the NADPH oxidase complex generates reactive oxygen species that lock the extracellular matrix in a continuous cycle of oxidative degradation, a process consistently replicated in ApoE⁻/⁻ animal models of atherosclerosis-driven aneurysmal expansion.[1]
| MOLECULAR MECHANISM
MMP/TIMP Imbalance: The proteolytic imbalance in AAA is so pronounced that MMP-9 levels in aneurysmal tissue can exceed those in normal aortic wall by more than tenfold, driving the fragmentation of elastin fibers that are essentially irreplaceable in the adult vasculature. Once elastin is destroyed, the structural loss is permanent. |
Thoracic Aortic Aneurysms: A Genetic–Structural Disease
TAA occupies fundamentally different biological territory. Rather than atherosclerotic inflammation, the dominant pathological process is intrinsic structural failure of the medial layer—what the 2022 ACC/AHA guidelines term “medial degeneration.”[5] This failure is frequently genetically encoded. Marfan syndrome, caused by mutations in the FBN1 gene encoding fibrillin-1, disrupts the microfibrillar network that anchors elastin within the media. Loeys-Dietz syndrome arises from mutations in TGF-β receptors (TGFBR1, TGFBR2) or their downstream effectors (SMAD2, SMAD3), producing a paradoxical overactivation of TGF-β signaling that drives disorganized fibrosis and progressive mechanical incompetence of the aortic root.[1]
What distinguishes heritable thoracic aortic disease from its abdominal counterpart is natural history: in these genetic conditions, catastrophic aortic events—including dissection and rupture—can occur at substantially smaller aortic diameters and at younger ages, making early identification and surveillance of affected individuals and their first-degree relatives a genuine lifesaving intervention.[6]
The Vasa Vasorum: Where the Aorta Dies from the Outside In
An increasingly recognized pathological mechanism bridges the worlds of atherosclerotic and structural aortic disease: the progressive dysfunction of the vasa vasorum—the intricate microvessel network embedded in the aortic adventitia, responsible for supplying oxygen and nutrients to the outer layers of the exceptionally thick aortic media.[7]
In patients with ascending aortic aneurysms, advanced imaging and histological studies reveal significant vasa vasorum remodeling alongside a detrimental downregulation of angiogenic signaling in the adventitia.[8] Histological evaluation of human aneurysmal tissue has identified obturating thrombi within the vasa vasorum itself—microvascular clots that sever the wall’s blood supply, producing chronic medial hypoxia, widespread smooth muscle cell necrosis, and profound mechanical brittleness.[9] The spontaneous rupture of pathologically neovascularized vasa vasorum vessels can precipitate an intramural hematoma—a pressurized blood collection within the medial layer that serves as an acute precursor to dissection or outward rupture.[10] The aorta, it turns out, is vulnerable from both its inner and outer surfaces simultaneously.
SECTION III
Physics Under Pressure: The Biomechanics of Athletic Exertion
Against this background of structural compromise, the question of athletic participation becomes a direct question of physics. The hemodynamic forces generated during exercise—systolic blood pressure, pulse pressure, cardiac output—impose physical tension on the aortic wall governed by Laplace’s Law, expressed as T = (P × R) / H, where wall tension (T) is a product of transmural pressure (P) and internal radius (R), divided by wall thickness (H). An aneurysmal aorta simultaneously has a larger radius and a thinned, degraded wall. Any exercise-induced increase in blood pressure therefore proportionally increases the tension applied to tissue that is already structurally compromised.[11]
An aneurysmal aorta has a larger radius and a thinner wall. Any rise in blood pressure proportionally amplifies tension in tissue already at the edge of mechanical failure.
Resistance Training and the Valsalva Paradox
Heavy strength training, powerlifting, and gymnastics share a critical biomechanical feature: intense voluntary muscular contractions almost invariably accompanied by the Valsalva maneuver—a forced exhalation against a closed glottis. This action drives profound, acute spikes in systemic arterial pressure, and has historically been viewed as the most dangerous form of exertion for any patient with aortic disease.[12]
Contemporary physiological research reveals a more nuanced picture. While absolute intra-arterial pressures can exceed 300 mmHg during a maximal lift, the concurrent Valsalva maneuver also dramatically increases intrathoracic pressure (ITP) and intracranial pressure (ICP). Research using continuous invasive monitoring—esophageal catheters and ventricular drains during graded lifting protocols—has demonstrated that the surge in ITP acts as an external counter-pressure on the intrathoracic vessels, such that actual transmural pressure across the ascending aorta may not rise as dramatically as absolute arterial pressure implies. Cerebrovascular transmural pressure has been shown to decrease by approximately 20 mmHg during the strain phase.[12]
The acute danger, however, arrives immediately upon releasing the Valsalva strain. The sudden drop in intrathoracic pressure generates violent backward wave reflections within the aorta, driving rapid, uncontrolled spikes in aortic systolic and pulse pressures. It is this rapid impedance shift—not the peak pressure during the lift itself—that is hypothesized to trigger intimal tears in vulnerable aortas.[12] High-intensity resistance training taken to muscular failure therefore remains a genuine, well-documented aortic risk.
Endurance Training: Volume Load and Aortic Mechanical Efficiency
Endurance modalities—long-distance running, cycling, rowing—impose a volume load rather than an acute pressure spike. During maximal aerobic exercise, cardiac output increases up to fivefold, and while mean arterial pressure rises modestly compared to weightlifting, the aorta must continuously accommodate massive stroke volumes at high heart rates.[13]
Landmark biomechanical work using cardiac MRI has directly quantified in-vivo Aortic Wall Stress (AWS) in patients with stable thoracic aortic disease during light-to-moderate aerobic exercise (3–5 METs). Peak ascending AWS ranged between 257 kPa and 323 kPa—approximately 70% to 73% below the experimentally established rupture threshold of 850–1,200 kPa for aneurysmal tissue.[14] Submaximal aerobic exercise, on current evidence, appears mechanically safe in terms of absolute wall stress.
Yet these studies identified an important nuance: individuals with thoracic aortic aneurysms exhibited AWS-to-cardiac-output slopes approximately 30% higher than age-matched controls. The diseased, stiffened aorta absorbs a disproportionate share of mechanical stress per unit of delivered blood flow—a physiological inefficiency that mandates careful, progressive scaling of any endurance program to prevent long-term matrix fatigue.[14]
| KEY FINDING — WALL STRESS DATA
At 3–5 METs of aerobic effort, peak aortic wall stress in patients with thoracic aortic disease reached 257–323 kPa—well below the 850–1,200 kPa rupture threshold. However, the diseased aorta generated ~30% more stress per unit of cardiac output than healthy controls, underscoring the importance of individualized intensity limits. |
SECTION IV
The Athlete’s Aorta: Adaptation or Disease?
Prolonged athletic training induces well-documented morphological adaptations across the cardiovascular system. While the “athlete’s heart” has been studied for decades, modern high-resolution imaging has revealed that the aorta itself undergoes measurable physiological remodeling in response to habitual hemodynamic loading, challenging the historical assumption that this great vessel is structurally static.[15]
A comprehensive meta-analysis of 9,464 elite athletes versus 2,637 healthy non-athlete controls confirmed that absolute aortic diameters at the sinuses of Valsalva are significantly larger in athletic populations—on average, 1.69 mm greater in young elite athletes under 35.[16] The clinical significance of this difference is magnified substantially with decades of athletic exposure. A landmark cross-sectional study of 442 masters endurance athletes—rowers and marathon runners aged 50–75 with at least ten years of intensive training post-age 40—found that 21% possessed aortic root diameters exceeding 40 mm, including 31% of male participants.[17] An equally striking 24% of this athletic cohort exhibited aortic dimensions more than two standard deviations above the general population mean.[18]
The pattern extends to power sports. Clinical evaluation of former professional American football players revealed that 29.6% harbored aortic diameters greater than 40 mm, versus just 8.6% in rigorously matched non-athlete controls.[15] This prevalence creates a genuine diagnostic challenge: at what point does a lifelong athlete’s enlarged aorta stop representing healthy adaptation and begin representing silent pathology?
Indexing: The Essential Correction
Because body size is the primary biological determinant of aortic diameter, indexing aortic measurements to body surface area (BSA) or height is a clinical imperative. When appropriately indexed, much of the absolute enlargement seen in large-framed athletes normalizes entirely—confirming proportional growth rather than pathological dilation.[16] A z-score greater than 2.0 after indexing shifts the diagnosis firmly toward an underlying aortopathy requiring full investigation. In young elite male athletes under 35, an absolute aortic dimension exceeding 42 mm mandates comprehensive workup including genetic screening and detailed family history for sudden cardiac death.[15]
SECTION V
Clinical Guidelines: From Prohibition to Precision
Clinical management of athletes with aortic disease has undergone a profound philosophical shift. Modern guidelines—anchored in the 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease[5] and operationalized for athletic populations by the 2025 ACC/AHA Scientific Statement on Clinical Considerations for Competitive Sports Participation[6]—have embraced a framework of Shared Decision-Making (SDM) that acknowledges both the risks of disease and the devastating consequences of enforced sedentarism.
Bicuspid Aortic Valve: A More Permissive Calculus
Bicuspid aortic valve (BAV) is the most common congenital cardiac defect globally, and approximately half of affected patients develop secondary ascending aortic dilation. The connective tissue vulnerability in BAV aortopathy, while real, is generally more localized and statistically less prone to rupture at small diameters than the aggressive syndromic heritable diseases. The 2025 ACC/AHA sports statement reflects this with a graduated, diameter-based participation framework.[6]
TABLE 1 — 2025 ACC/AHA SPORTS PARTICIPATION RECOMMENDATIONS: BICUSPID AORTIC VALVE
| AORTIC DIAMETER (BAV) | RECOMMENDATION | KEY CONSIDERATIONS |
| ≤ 42 mm | Cleared for participation | Low risk; all athletic disciplines generally permitted with standard surveillance |
| 43–44 mm | Consider via Shared Decision-Making | Borderline zone; requires detailed informed consent and frequent echocardiographic monitoring |
| ≥ 45 mm | Generally restricted | Dissection risk likely outweighs competitive participation benefit; highly individualized exceptions only |
| ≥ 50 mm (masters) | Surgical correction recommended before clearance | Prophylactic ascending aortic replacement strongly considered before resuming competitive exertion |
The 2024 ESC Guidelines for Peripheral Arterial and Aortic Diseases further specify that when a BAV patient requires cardiac surgery for valve dysfunction, the threshold for concomitant ascending aortic replacement drops to ≥ 45 mm—significantly lower than the ≥ 55 mm threshold applied to patients with normal tricuspid valves and no genetic syndrome.[2]
Heritable Thoracic Aortic Disease: A More Restrictive Calculus
Patients with confirmed syndromic heritable thoracic aortic disease (HTAD)—Marfan syndrome, Loeys-Dietz syndrome, Vascular Ehlers-Danlos syndrome—face a substantially more guarded prognosis. The systemic vulnerability of the vascular tree in these conditions means that catastrophic events can occur at smaller diameters and with less warning than in BAV or degenerative aortic disease. Participation in competitive high-intensity sports, sustained isometric exertion, and high-impact collision sports is universally discouraged.[19]
Complete enforced sedentarism is, however, also recognized as harmful—both metabolically and psychologically. Moderate recreational aerobic exercise is strongly encouraged to control resting blood pressure, optimize endothelial function, and preserve overall cardiovascular efficiency, even in the highest-risk patients.[15]
SECTION VI
Safe Exercise Prescription: Evidence-Based Protocols
For patients definitively diagnosed with aortic aneurysm who have been cleared for non-competitive physical activity, establishing objective hemodynamic safety boundaries is critical. The evidence base now clearly supports that low-to-moderate intensity aerobic exercise does not accelerate longitudinal aneurysm growth rates and provides substantial metabolic benefit.[20]
A landmark clinical trial published in Frontiers in Sports and Active Living established a reproducible, moderate-intensity circuit training protocol with rigorous ambulatory blood pressure monitoring for patients with thoracic aortic aneurysm and post-dissection status.[21] The protocol defined four critical safety parameters:
| EVIDENCE-BASED SAFETY THRESHOLDS
1. Baseline exclusion: Do not initiate exercise if resting systolic blood pressure exceeds 160 mmHg. Pharmacological control is a prerequisite.2. Intensity monitoring: Use the Borg CR-10 perceived exertion scale to target “moderate” effort—a non-invasive proxy that reliably limits hidden hemodynamic spikes.3. Hemodynamic ceilings: Aim for exertional systolic BP below 160 mmHg during training. Mandatory termination if systolic BP exceeds 230 mmHg, diastolic BP exceeds 120 mmHg, or chest discomfort develops.4. Isometric restrictions: Limit resistance exercise to below 40–50% of one-repetition maximum. Continuous breathing throughout—the Valsalva maneuver must be avoided absolutely. |
TABLE 2 — STANDARDIZED MODERATE-INTENSITY PROTOCOL FOR AORTIC DISEASE PATIENTS
| EXERCISE | PROTOCOL PARAMETERS |
| Hand Grips (isometric) | 40% of maximal voluntary exertion, dominant hand |
| Leg Raises (dynamic core) | Supine position; heels elevated 6 inches above ground |
| Bicep Curls (isotonic) | 5–10 lb dumbbells; continuous breathing throughout |
| Stationary Cycling (aerobic) | 80–100 Watts target output |
| Wall Sits (isometric) | 90° angle, back to thighs; continuous nasal breathing |
A clinically significant observation from this trial: patients reporting higher habitual weekly activity levels demonstrated significantly lower exertional systolic blood pressure spikes during formal testing—a direct, measurable anti-hypertensive effect of sustained baseline fitness.[21] Clinicians must also account for pharmacological context; patients on beta-blockers exhibited substantially blunted maximum exercise heart rates (approximately 92.8 bpm versus 112.0 bpm in unmedicated patients), requiring modification of any heart-rate-based exercise targets.[21]
SECTION VII
Recognizing Catastrophe: Warning Signs of Acute Aortic Syndromes
Even with scrupulous adherence to activity modification and surveillance protocols, acute aortic syndromes can and do occur in athletic populations. Recognizing their presentation can mean the difference between survival and death—for the athlete, for coaches on the sideline, and for first responders.
Aortic dissection announces itself with a signature unlike almost any other cardiovascular emergency: pain that is maximal at its precise moment of onset, rather than building gradually as ischemic chest pain does. This tearing or ripping pain is typically central in the chest, frequently radiating to the suprasternal notch or straight through to the interscapular back, following the anatomical trajectory of the tear.[22] A sudden syncopal event during competition or in the weight room—occurring without clear vasovagal trigger—must always raise the index of suspicion for a hemodynamic collapse secondary to aortic catastrophe.[22]
Physical examination may reveal three critical diagnostic clues: the acute development of a new diastolic murmur indicating aortic valve regurgitation as the dissection flap extends into the root; asymmetrical pulse deficits or blood pressure discrepancies between limbs, reflecting branch vessel obstruction by the advancing flap; and the classic triad of cardiac tamponade—refractory hypotension, elevated jugular venous pressure, and muffled heart sounds—signaling retrograde hemorrhage into the pericardial sac.[23]
Any of these findings demands immediate cessation of activity, aggressive pharmacological blood pressure and heart rate reduction (anti-impulse therapy), and emergent transfer to a high-volume quaternary center with a dedicated Multidisciplinary Aortic Team.[5]
CONCLUSION
Precision Over Prohibition
The management of athletes with aortic disease demands a clinical synthesis of molecular biology, hemodynamic physics, and individualized risk assessment that was not possible—and in some cases not attempted—a decade ago. The fundamental pathological distinction between the atherosclerosis-driven inflammatory destruction of AAA and the genetic, structurally driven medial degeneration of TAA must anchor every clinical decision. The biomechanical evidence is increasingly clear: moderate aerobic exercise is both safe and beneficial, while high-intensity resistance training with uncontrolled Valsalva maneuver represents genuine, quantifiable risk.
The “athlete’s aorta” phenomenon demands that every enlarged vessel in a high-level athlete be rigorously indexed to body surface area before pathological significance is assigned or withheld. The graduated, diameter-based participation frameworks of the 2025 ACC/AHA sports guidelines, applied through Shared Decision-Making, represent a mature evidence-based evolution away from blanket prohibition toward individualized precision.
The goal is not the elimination of physical activity—it is the intelligent prescription of it. For a patient with a vulnerable aorta, the right exercise, at the right intensity, with the right monitoring, is not merely permitted. It is protective.
References
- Nadel J, Rodríguez-Palomares J, Phinikaridou A, Prieto C, Masci PG, Botnar R. The future of cardiovascular magnetic resonance imaging in thoracic aortopathy: blueprint for the paradigm shift to improve management. J Cardiovasc Magn Reson. 2025;27(1):101865. doi:10.1016/j.jocmr.2025.101865
- Mazzolai L, Teixido-Tura G, Lanzi S, et al. 2024 ESC Guidelines for the management of peripheral arterial and aortic diseases. Eur Heart J. 2024;45(36):3538-3700. doi:10.1093/eurheartj/ehae179
- Golledge J, Norman PE. Atherosclerosis and abdominal aortic aneurysm: cause, response, or common risk factors?. Arterioscler Thromb Vasc Biol. 2010;30(6):1075-1077. doi:10.1161/ATVBAHA.110.206573
- Sweeting MJ, Balm R, Desgranges P, Ulug P, Powell JT; Ruptured Aneurysm Trialists. Individual-patient meta-analysis of three randomized trials comparing endovascular versus open repair for ruptured abdominal aortic aneurysm. Br J Surg. 2015;102(10):1229-1239. doi:10.1002/bjs.9852
- Isselbacher EM, Preventza O, Hamilton Black J 3rd, et al. 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease: A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2022;146(24):e334-e482. doi:10.1161/CIR.0000000000001106
- Kim JH, Baggish AL, Levine BD, et al. Clinical Considerations for Competitive Sports Participation for Athletes With Cardiovascular Abnormalities: A Scientific Statement From the American Heart Association and American College of Cardiology. J Am Coll Cardiol. 2025;85(10):1059-1108. doi:10.1016/j.jacc.2024.12.025
- Xu J, Lu X, Shi GP. Vasa vasorum in atherosclerosis and clinical significance. Int J Mol Sci. 2015;16(5):11574-11608. Published 2015 May 20. doi:10.3390/ijms160511574
- Billaud M, Hill JC, Richards TD, Gleason TG, Phillippi JA. Medial Hypoxia and Adventitial Vasa Vasorum Remodeling in Human Ascending Aortic Aneurysm. Front Cardiovasc Med. 2018;5:124. Published 2018 Sep 17. doi:10.3389/fcvm.2018.00124
- Chumachenko PV, Ivanova AG, Bagheri Ekta M, et al. Condition “Vasa Vasorum” in Patients with Thoracic Aortic Aneurysm. J Clin Med. 2023;12(10):3578. Published 2023 May 20. doi:10.3390/jcm12103578
- Baikoussis NG, Apostolakis EE, Papakonstantinou NA, et al. The implication of vasa vasorum in surgical diseases of the aorta. Eur J Cardiothorac Surg. 2011;40(2):412-417. doi:10.1016/j.ejcts.2010.11.045
- Haykowsky MJ, Skow RJ, Foulkes SJ, et al. Aorta Wall Stress during Exercise in Patients with an Ascending Thoracic Aortic Aneurysm: Insights from a Case Series. Aorta (Stamford). 2024;12(5):108-116. doi:10.1055/a-2558-4266
- Wakeham DJ, Pierce GL, Heffernan KS. Effect of Acute Resistance Exercise and Resistance Exercise Training on Central Pulsatile Hemodynamics and Large Artery Stiffness: Part I. Pulse (Basel). 2025;13(1):31-44. Published 2025 Feb 7. doi:10.1159/000543313
- Horn P, Ostadal P, Ostadal B. Rowing increases stroke volume and cardiac output to a greater extent than cycling. Physiol Res. 2015;64(2):203-207. doi:10.33549/physiolres.932853
- Skow RJ, Foulkes SJ, Thompson RB, et al. Acute effects of aerobic exercise on aortic wall stress in thoracic aortic disease. Am J Physiol Heart Circ Physiol. 2026;330(2):H338-H347. doi:10.1152/ajpheart.00829.2025
- Iskandar A, Thompson PD. Diseases of the Aorta in Elite Athletes. Clin Sports Med. 2015;34(3):461-472. doi:10.1016/j.csm.2015.03.001
- Rodríguez-Palomares JF, Oliveró R, Teixidó-Tura G. Understanding Aortic Enlargement in Elite Athletes: A Physiological Adaptation or Pathological Concern?. Eur J Prev Cardiol. Published online February 19, 2025. doi:10.1093/eurjpc/zwaf082
- Carbone A, Monda E, Ferrara F, et al. Aortic Dimension in Elite Athletes: Updated Systematic Review and Meta-Analysis. Eur J Prev Cardiol. Published online November 26, 2024. doi:10.1093/eurjpc/zwae385
- Churchill TW, Baggish AL. Cardiovascular Care of Masters Athletes. J Cardiovasc Transl Res. 2020;13(3):313-321. doi:10.1007/s12265-020-09987-2
- Braverman AC, Harris KM, Kovacs RJ, Maron BJ; American Heart Association Electrocardiography and Arrhythmias Committee of Council on Clinical Cardiology, Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and American College of Cardiology. Eligibility and Disqualification Recommendations for Competitive Athletes With Cardiovascular Abnormalities: Task Force 7: Aortic Diseases, Including Marfan Syndrome: A Scientific Statement From the American Heart Association and American College of Cardiology. Circulation. 2015;132(22):e303-e309. doi:10.1161/CIR.0000000000000243
- Stiefel M, Brito da Silva H, Schmied CM, Niederseer D. Exercise, Sports, and Cardiac Rehabilitation Recommendations in Patients with Aortic Aneurysms and Post-Aortic Repair: A Review of the Literature. J Cardiovasc Dev Dis. 2024;11(12):379. Published 2024 Nov 27. doi:10.3390/jcdd11120379
- Li J, Boyd A, Huang M, Berookhim J, Prakash SK. Safety of exercise for adults with thoracic aortic aneurysms and dissections. Front Sports Act Living. 2022;4:888534. Published 2022 Aug 22. doi:10.3389/fspor.2022.888534
- Singh B, Treece JM, Murtaza G, Bhatheja S, Lavine SJ, Paul TK. Aortic Dissection in a Healthy Male Athlete: A Unique Case with Comprehensive Literature Review. Case Rep Cardiol. 2016;2016:6460386. doi:10.1155/2016/6460386
- Gurevitz M, Weinberger A, Miller D. Aortic Root Aneurysm in an Extreme Athlete. Cureus. 2022;14(7):e26661. Published 2022 Jul 8. doi:10.7759/cureus.26661