Not all fat is the same — and where your body stores it turns out to matter a lot more than how much you weigh.
Have you ever noticed that two people can weigh exactly the same and be completely different when it comes to their health? That’s because weight alone doesn’t tell the whole story. Scientists have discovered that where your body stores fat is what really matters — and fat stored deep in your belly, called visceral fat, is in a league of its own when it comes to health risks.
For a long time, scientists thought fat was just a boring storage tank — like a spare gas tank your body uses when it runs low on energy. But in 1994, researchers made a surprising discovery: fat actually talks. It sends chemical messages to your brain and other organs, acting like its own little hormone factory. This changed everything about how we understand obesity and metabolic health.
KEY IDEA There are two main types of body fat. Subcutaneous fat sits just under your skin — you can pinch it. Visceral fat hides deep inside your belly, packed around your organs like the liver, stomach, and intestines. You can’t see or feel it from the outside.
The plumbing problem
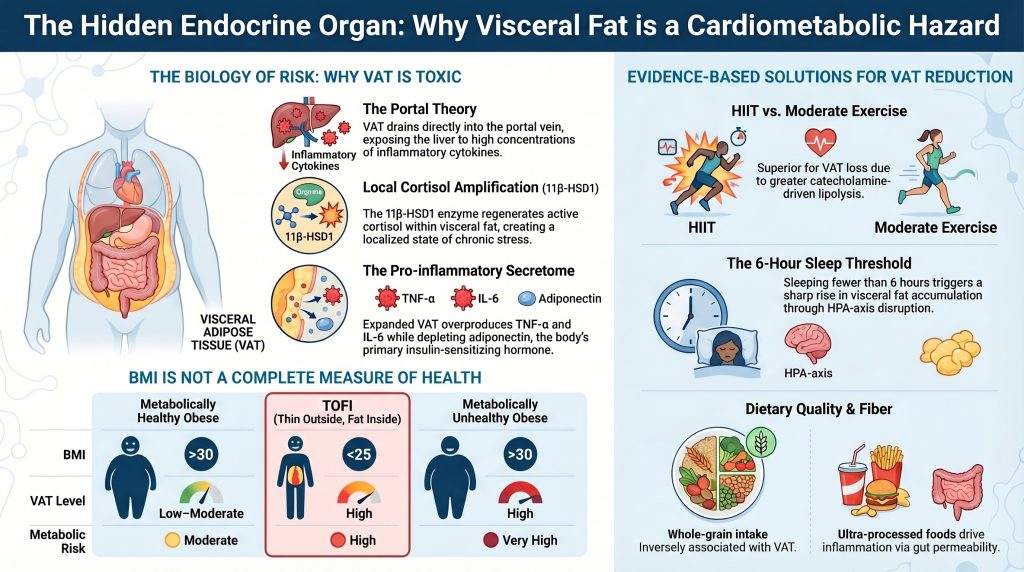
So why is visceral fat the troublemaker? It comes down to plumbing. Visceral fat sits right next to a major blood highway called the portal vein, which connects directly to your liver. When that fat releases chemicals and fatty acids into your blood, your liver gets hit first and hardest. It’s like living next door to a factory that dumps pollution directly into your water supply. The result? Your liver starts malfunctioning — producing too much sugar and too much fat in your blood — which sets the stage for diabetes and heart disease.
Visceral fat is also a drama queen when it comes to inflammation. In people who carry a lot of it, fat cells become stressed and swollen. They start releasing alarm chemicals called cytokines — basically distress signals that put your whole immune system on edge. This low-grade inflammation is like having a tiny, slow-burning fire inside your body at all times. Over time, that constant inflammation damages blood vessels and increases the risk of heart attacks and strokes.
Stress makes it worse
Here’s another twist: stress makes things worse. When you’re stressed, your body releases a hormone called cortisol. Cortisol is useful in short bursts — it helps you handle emergencies. But when stress becomes chronic, cortisol levels stay high, and your body responds by storing more fat in your belly. Basically, stress tells your body to build up visceral fat reserves, which then causes more inflammation, which can cause more health problems. It’s a frustrating cycle.
The “Thin Outside, Fat Inside” effect
One of the strangest discoveries in this area is the so-called “TOFI” effect — Thin Outside, Fat Inside. Some people look perfectly healthy and have a normal body weight, but they’re carrying surprisingly high amounts of visceral fat around their organs. Their BMI — the number doctors often use to measure healthy weight — looks fine, but the fat that really matters is hidden from view. This is one reason scientists say BMI alone is a poor way to measure health.
Sleep also plays a surprising role. Studies show that people who sleep fewer than six hours a night tend to build up more visceral fat over time. Poor sleep throws off hormone levels — including cortisol, and a hunger hormone called ghrelin — making people eat more and store more fat in exactly the wrong place.
THE GOOD NEWS Visceral fat is actually one of the more responsive types of fat to lifestyle changes. Diet, exercise, stress management, and better sleep can all make a real difference — even when the scale barely moves.
What you can do about it
When it comes to exercise, research shows that intense workouts — where you push hard for short bursts, then rest, then repeat — are especially effective at burning visceral fat. Scientists call this HIIT (High-Intensity Interval Training). Even steady cardio like walking or jogging helps. The key is simply moving consistently.
Diet matters too. Whole grains, vegetables, fruit, and lean protein help keep visceral fat in check. Ultra-processed foods — the ones that come in shiny packages and have ten ingredients you can’t pronounce — seem to do the opposite, encouraging fat to pile up in the belly. Cutting back on sugary drinks is also one of the highest-impact changes you can make.
The bottom line? Your bathroom scale can’t tell you everything your doctor might want to know. The fat you can’t see — tucked deep inside your abdomen — is quietly influencing your risk for heart disease, diabetes, and other serious conditions. The science of visceral fat is a reminder that when it comes to health, what’s on the inside really does count.
Based on peer-reviewed research in endocrinology, metabolic medicine, and exercise science. Always consult a healthcare provider for personal medical advice.
DEEP DIVE
The Pathophysiology of Visceral Adiposity
Evolutionary Origins, Endocrine Signaling, and Cardiometabolic Implications
Comprehensive Review | April 2026
ABSTRACT
White adipose tissue (WAT) has undergone a substantial conceptual shift over the past three decades, from a passive reservoir of triglycerides to a metabolically active endocrine organ that plays a central role in systemic homeostasis. Within this framework, visceral adipose tissue (VAT) is associated with a disproportionately high cardiometabolic risk profile relative to total adiposity. This is largely attributable to three defining features: its direct drainage into the portal circulation, its enhanced capacity for local glucocorticoid activation via 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1), and its pro-inflammatory secretory phenotype. Together, these characteristics contribute to insulin resistance, hepatic steatosis, dyslipidemia, and systemic inflammation. This review synthesizes current peer-reviewed evidence on the evolutionary context of visceral fat, its endocrine and paracrine signaling mechanisms, its contribution to cardiometabolic disease, and evidence-based strategies for its reduction, while distinguishing established physiological mechanisms from emerging or theoretical models.
-
Introduction
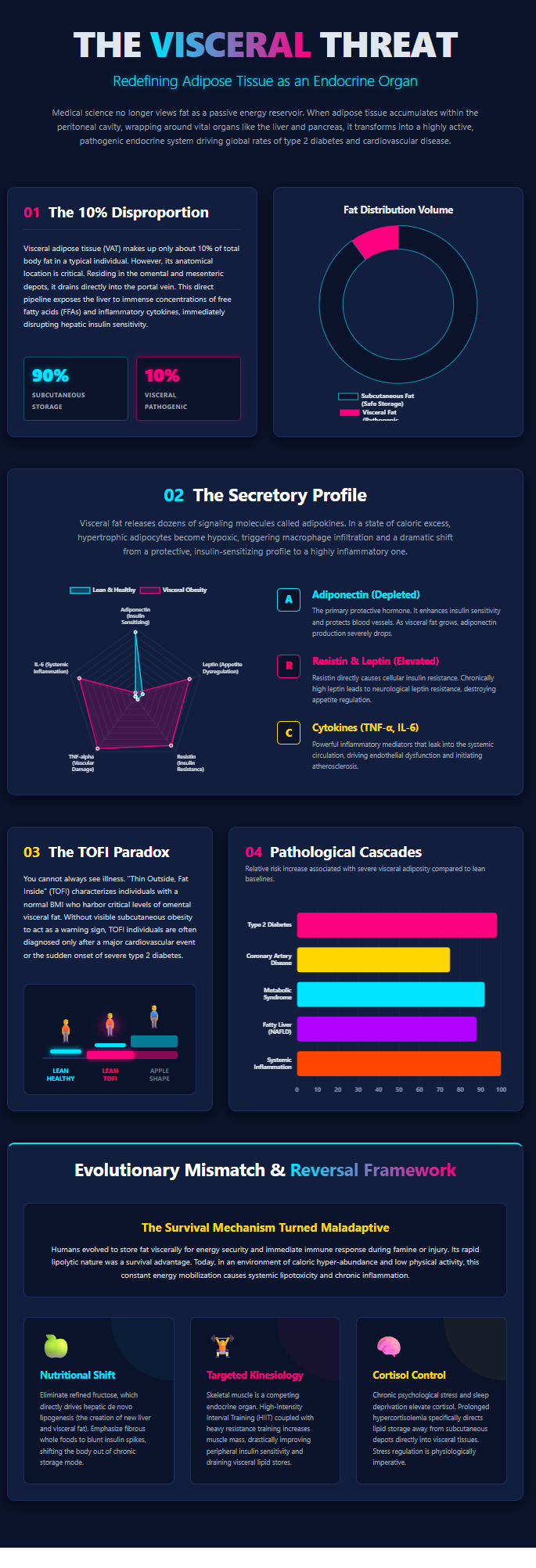
The discovery of leptin in 1994 provided the first clear evidence that adipose tissue functions as an endocrine organ capable of communicating with the central nervous system to regulate energy balance [1]. Since then, adipose tissue has been recognized as a dynamic regulator of metabolic, inflammatory, and endocrine processes. Importantly, adipose tissue is not functionally homogeneous. The distribution of fat—particularly the accumulation of visceral adipose tissue within the intra-abdominal cavity—has been shown to be a stronger predictor of cardiometabolic morbidity and mortality than total fat mass or body mass index (BMI) [4], [16].
Visceral adipose tissue differs from subcutaneous adipose tissue (SAT) in several critical respects, including cellular composition, vascular drainage, and endocrine activity [6]. While SAT primarily serves as a relatively inert energy storage depot, VAT is more metabolically active and exhibits a distinct secretory profile characterized by increased production of pro-inflammatory mediators. Proteomic studies suggest that adipose tissue secretes hundreds of bioactive molecules, collectively referred to as adipokines, which regulate systemic metabolism and immune function [2], [6]. In states of visceral obesity, the balance of these signals shifts toward pathways that promote insulin resistance, endothelial dysfunction, and atherogenesis.
-
Evolutionary Perspectives and the Adipo-Immune Function of Visceral Fat
2.1 Evolutionary Models of Fat Distribution
The distribution of body fat is a regulated, heritable trait influenced by evolutionary pressures. The “thrifty gene” hypothesis posits that the ability to efficiently store energy in adipose tissue conferred a survival advantage during periods of intermittent famine [9]. However, this model does not fully account for the substantial inter-individual variability observed in modern populations.
The “drifty gene” hypothesis provides an alternative explanation, suggesting that genetic variation in adiposity accumulated through neutral drift once predation pressure—an evolutionary constraint on excess body mass—was reduced [9]. While this model explains variability, it does not address depot-specific fat distribution.
More recent frameworks propose that visceral adipose tissue may have had adaptive immune functions. The “VAT prioritization” hypothesis suggests that preferential expansion of visceral fat could have supported host defense by providing both energy reserves and immune signaling capacity during infection and malnutrition [12]. Although biologically plausible, this hypothesis remains speculative and should be interpreted as an emerging conceptual model rather than established consensus.
2.2 The Omentum as an Immunological Structure
The omentum, a peritoneal structure overlying the abdominal organs, contains specialized immune aggregates known as fat-associated lymphoid clusters (FALCs) and milky spots [12]. These structures contribute to immune surveillance and play a role in the containment of intra-abdominal infections.
Visceral adipocytes are capable of interacting with immune pathways through cytokine signaling and lipid provision for immune cell function. In conditions of chronic caloric excess, alterations in gut microbiota composition and increased intestinal permeability have been associated with elevated circulating lipopolysaccharide (LPS) levels, a phenomenon referred to as metabolic endotoxemia [12]. This process has been implicated in low-grade systemic inflammation; however, it is best understood as one of several contributing mechanisms rather than a universally causal pathway [5], [12].
-
The Endocrine Architecture of Visceral Adipose Tissue
3.1 Leptin and Adiponectin
Leptin, produced predominantly by adipocytes, serves as a key regulator of energy balance by signaling nutritional status to the hypothalamus [2], [3]. It suppresses appetite and promotes energy expenditure through activation of JAK2/STAT3 signaling pathways. In obesity, circulating leptin levels are elevated, yet this is often accompanied by central leptin resistance, partially mediated by increased expression of suppressor of cytokine signaling-3 (SOCS3), which attenuates downstream signaling [3].
Adiponectin exerts opposing metabolic effects, enhancing insulin sensitivity and promoting fatty acid oxidation through activation of AMP-activated protein kinase (AMPK) [1], [3]. Unlike leptin, adiponectin levels are inversely correlated with adiposity, particularly visceral fat accumulation. Reduced adiponectin concentrations are consistently associated with increased cardiometabolic risk and may serve as an early biomarker of metabolic dysfunction [6].
3.2 Pro-inflammatory Adipokines and Cytokines
A defining feature of visceral adiposity is the increased production of pro-inflammatory mediators, including tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) [2], [5]. In hypertrophic adipose tissue, local hypoxia promotes the recruitment and activation of macrophages, particularly the pro-inflammatory M1 phenotype.
TNF-α contributes to insulin resistance by inducing serine phosphorylation of insulin receptor substrate proteins, thereby impairing insulin signaling pathways [5]. IL-6, which is released in significant quantities from visceral depots, enters the portal circulation and stimulates hepatic production of C-reactive protein (CRP), a key marker and mediator of systemic inflammation [15].
Additional adipokines, such as resistin and retinol-binding protein 4 (RBP4), have been associated with impaired glucose metabolism and insulin resistance, although their precise roles in humans remain an area of ongoing investigation [2].
-
Anatomical Hazard: The Portal Theory and Hepatic Lipotoxicity
4.1 The Portal Theory
Visceral adipose tissue is uniquely positioned anatomically due to its direct drainage into the portal vein, which delivers blood to the liver before it enters the systemic circulation [13]. This arrangement forms the basis of the “portal theory,” which proposes that the liver is exposed to disproportionately high concentrations of free fatty acids (FFAs) and pro-inflammatory cytokines released by visceral fat.
As visceral adipose tissue becomes insulin-resistant, lipolysis increases, resulting in elevated FFA flux into the portal circulation. These FFAs contribute to hepatic insulin resistance by promoting gluconeogenesis and increasing very-low-density lipoprotein (VLDL) production [4], [5]. This process contributes to hyperglycemia and dyslipidemia, key components of metabolic syndrome.
Experimental studies support this mechanism. Selective transplantation of adipose tissue into the portal circulation in animal models induces hepatic insulin resistance, whereas transplantation into the systemic circulation does not produce the same effect [20].
4.2 Ectopic Fat and the Expandability Hypothesis
When subcutaneous adipose tissue reaches its storage capacity, excess lipids are deposited in ectopic locations, including the liver, pancreas, skeletal muscle, and myocardium [7]. This process contributes to lipotoxicity and organ dysfunction.
The “expandability hypothesis” suggests that the capacity of subcutaneous fat to safely store lipids varies among individuals, influencing susceptibility to ectopic fat accumulation and metabolic disease [7]. Although widely supported, this framework continues to evolve as new data emerge.
-
The TOFI Phenotype and Metabolic Heterogeneity
The “Thin Outside, Fat Inside” (TOFI) phenotype describes individuals with normal BMI but elevated visceral fat and increased cardiometabolic risk [29], [31]. These individuals may exhibit insulin resistance, dyslipidemia, and increased hepatic fat despite appearing lean.
Imaging studies demonstrate that TOFI individuals often have reduced subcutaneous fat alongside increased visceral and intrahepatic fat stores [30]. This highlights the limitation of BMI as a sole indicator of metabolic health and underscores the importance of assessing fat distribution.
-
Chronic Stress, the HPA Axis, and Visceral Fat Accumulation
6.1 Cortisol and the 11β-HSD1 Amplification Loop
Chronic psychological and physiological stress activates the hypothalamic–pituitary–adrenal (HPA) axis, leading to sustained elevations in circulating cortisol [38]. Cortisol influences adipose tissue distribution and metabolism through several mechanisms. It promotes the differentiation of preadipocytes into mature adipocytes, enhances lipoprotein lipase (LPL) activity in adipose tissue, and facilitates lipid storage, particularly in visceral depots where glucocorticoid receptor expression is relatively high [39], [41].
In addition to systemic effects, cortisol activity is amplified locally within adipose tissue by 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1), which converts inactive cortisone into active cortisol [42]. Expression of 11β-HSD1 is higher in visceral adipose tissue than in subcutaneous depots in many experimental contexts, contributing to a localized environment that may resemble features of Cushing’s syndrome at the tissue level. It should be noted, however, that the depot-specific predominance of 11β-HSD1 has not been uniformly confirmed across all human studies, and its precise regulatory role continues to be investigated [42].
Experimental models strongly support the role of 11β-HSD1 in metabolic dysfunction. Transgenic mice overexpressing this enzyme selectively in adipose tissue develop visceral obesity, insulin resistance, dyslipidemia, and hypertension [41]–[43]. These findings suggest that local glucocorticoid amplification contributes significantly to the metabolic consequences of visceral adiposity.
6.2 Sleep Deprivation and Visceral Adiposity
Sleep duration is an important modulator of metabolic health. Short sleep duration, typically defined as fewer than six hours per night, has been associated with increased visceral adiposity and elevated Visceral Adiposity Index (VAI) scores [36], [46]. Sleep restriction disrupts circadian regulation of the HPA axis, leading to elevated evening cortisol levels.
In addition to cortisol dysregulation, sleep deprivation alters appetite-regulating hormones, including decreased leptin and increased ghrelin levels, which may promote increased caloric intake and weight gain [45]. Observational studies, including analyses of National Health and Nutrition Examination Survey (NHANES) data, suggest a non-linear relationship between sleep duration and visceral adiposity, with risk increasing significantly below approximately six hours per night [36].
Longitudinal data indicate that improvements in sleep duration are associated with reduced rates of visceral fat accumulation over time [49]. These findings support the inclusion of sleep optimization as part of comprehensive strategies to mitigate cardiometabolic risk.
-
Cardiovascular Implications and the Atherosclerosis Cascade
Visceral adiposity contributes to cardiovascular disease through a combination of metabolic, inflammatory, and hemodynamic mechanisms [37]. Increased visceral fat mass is associated with elevated circulating levels of pro-inflammatory cytokines, which promote endothelial dysfunction and vascular inflammation.
One key mediator is plasminogen activator inhibitor-1 (PAI-1), which is produced in increased quantities by visceral adipose tissue [19]. Elevated PAI-1 levels impair fibrinolysis, contributing to a prothrombotic state and increasing the risk of coronary artery disease.
Interleukin-6 (IL-6) released from visceral fat enters the portal circulation and stimulates hepatic production of C-reactive protein (CRP), which plays a role in endothelial activation and atherogenesis [14]. These processes contribute to the development of atherosclerotic plaques and vascular stiffness.
Epicardial adipose tissue, a visceral fat depot surrounding the heart, may exert local effects on myocardial function. Increased epicardial fat thickness has been associated with impaired coronary microcirculation, diastolic dysfunction, and arrhythmogenesis [14]. These findings highlight both systemic and localized cardiovascular effects of visceral adiposity.
-
Evidence-Based Interventions for Visceral Fat Reduction
8.1 Nutritional Strategies
Dietary composition plays a significant role in the regulation of visceral fat accumulation. Diets rich in whole grains, fiber, fruits, and vegetables are associated with lower visceral fat levels, while diets high in refined carbohydrates and sugar-sweetened beverages are associated with increased adiposity [17], [51].
Consumption of ultra-processed foods has been associated with increased energy intake, weight gain, and metabolic dysfunction [53], [54]. These foods may influence visceral adiposity through multiple pathways, including alterations in gut microbiota composition and increased intestinal permeability. While these mechanisms are supported by emerging evidence, their relative contribution remains an area of ongoing investigation.
Dietary patterns that emphasize minimally processed foods and high nutrient density are consistently associated with improved metabolic outcomes and reduced visceral fat accumulation.
8.2 Exercise: HIIT versus Moderate-Intensity Continuous Training
Physical activity is a key intervention for reducing visceral fat. Both high-intensity interval training (HIIT) and moderate-intensity continuous training (MICT) have been shown to reduce visceral adiposity [55], [58].
Meta-analyses indicate that HIIT may produce comparable or modestly greater reductions in visceral fat compared to MICT, although differences are not universally consistent across studies [55]. Proposed mechanisms include increased catecholamine release, which stimulates lipolysis, and elevated post-exercise oxygen consumption (EPOC), which enhances energy expenditure following exercise [59].
HIIT is also associated with improvements in cardiorespiratory fitness and insulin sensitivity. However, adherence, safety, and individual preference should be considered when prescribing exercise interventions.
8.3 Stress Management and Sleep Optimization
Given the role of HPA axis activation in visceral fat accumulation, stress management represents an important component of intervention strategies. Techniques such as mindfulness-based stress reduction (MBSR), relaxation training, and behavioral therapy have been associated with improvements in metabolic outcomes [63].
Sleep optimization is similarly important. Adequate sleep duration and quality are associated with improved hormonal regulation and reduced risk of visceral fat accumulation [49]. Interventions targeting sleep hygiene may therefore contribute to long-term cardiometabolic health.
-
Diagnostic Modalities and Clinical Assessment
Accurate assessment of visceral adiposity is essential for risk stratification and monitoring of treatment response. Imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) provide direct quantification of visceral fat.
CT imaging at the L4/L5 vertebral level is commonly considered a reference standard due to its ability to differentiate adipose tissue based on radiodensity [65]. However, its use is limited by exposure to ionizing radiation.
MRI offers a radiation-free alternative and allows for detailed assessment of fat distribution, including hepatic and pancreatic fat content [27], [66]. Despite its advantages, cost and accessibility may limit routine clinical use.
In clinical practice, surrogate indices such as the Metabolic Score for Visceral Fat (METS-VF) and the Visceral Adiposity Index (VAI) provide practical tools for estimating visceral fat–related risk [35]. These indices integrate anthropometric and metabolic variables and have demonstrated predictive value for cardiometabolic outcomes.
-
Conclusion
Visceral adiposity is a major contributor to cardiometabolic disease through a complex interplay of endocrine, inflammatory, and metabolic mechanisms. Key pathways include portal delivery of free fatty acids and cytokines to the liver, local amplification of glucocorticoid activity via 11β-HSD1, and the secretion of pro-inflammatory adipokines.
While several mechanisms underlying the pathological effects of visceral fat are well established, others—such as interactions between the gut microbiome and adipose tissue—remain areas of active investigation. Importantly, fat distribution rather than total fat mass appears to be a critical determinant of metabolic risk.
Effective management of visceral adiposity requires a multifaceted approach that includes dietary modification, physical activity, stress reduction, and sleep optimization. Continued research is needed to refine diagnostic tools and develop targeted interventions that address the underlying biological mechanisms of visceral fat accumulation.
References
- Kershaw EE, Flier JS. Adipose tissue as an endocrine organ. J Clin Endocrinol Metab. 2004;89(6):2548-2556. doi:10.1210/jc.2004-0395
- Trayhurn P, Wood IS. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr. 2004;92(3):347-355. doi:10.1079/bjn20041213
- Frühbeck G, Gómez-Ambrosi J, Muruzábal FJ, Burrell MA. The adipocyte: a model for integration of endocrine and metabolic signaling in energy metabolism regulation. Am J Physiol Endocrinol Metab. 2001;280(6):E827-E847. doi:10.1152/ajpendo.2001.280.6.E827
- Dubé MC, Lemieux S, Piché ME, et al. The contribution of visceral adiposity and mid-thigh fat-rich muscle to the metabolic profile in postmenopausal women. Obesity (Silver Spring). 2011;19(5):953-959. doi:10.1038/oby.2010.348
- Vicardi M, Farzaneh-Far A, Fava C, Dalle Carbonare L, Romano S. Epicardial and Visceral Adipose Tissue and Global Longitudinal Strain: A Review of Cardiac Imaging Insights in Subclinical Myocardial Dysfunction. Nutrients. 2026;18(6):1009. Published 2026 Mar 23. doi:10.3390/nu18061009
- Caputo T, Gilardi F, Desvergne B. From chronic overnutrition to metaflammation and insulin resistance: adipose tissue and liver contributions. FEBS Lett. 2017;591(19):3061-3088. doi:10.1002/1873-3468.12742
- Virtue S, Vidal-Puig A. Adipose tissue expandability, lipotoxicity and the Metabolic Syndrome–an allostatic perspective. Biochim Biophys Acta. 2010;1801(3):338-349. doi:10.1016/j.bbalip.2009.12.006
- Speakman JR, O’Rahilly S. Fat: an evolving issue. Dis Model Mech. 2012;5(5):569-573. doi:10.1242/dmm.010553
- Mraz M, Haluzik M. The role of adipose tissue immune cells in obesity and low-grade inflammation. J Endocrinol. 2014;222(3):R113-R127. doi:10.1530/JOE-14-0283
- Kabir M, Catalano KJ, Ananthnarayan S, et al. Molecular evidence supporting the portal theory: a causative link between visceral adiposity and hepatic insulin resistance. Am J Physiol Endocrinol Metab. 2005;288(2):E454-E461. doi:10.1152/ajpendo.00203.2004
- Iacobellis G. Epicardial adipose tissue in contemporary cardiology. Nat Rev Cardiol. 2022;19(9):593-606. doi:10.1038/s41569-022-00679-9
- Piché ME, Lemieux S, Weisnagel SJ, Corneau L, Nadeau A, Bergeron J. Relation of high-sensitivity C-reactive protein, interleukin-6, tumor necrosis factor-alpha, and fibrinogen to abdominal adipose tissue, blood pressure, and cholesterol and triglyceride levels in healthy postmenopausal women. Am J Cardiol. 2005;96(1):92-97. doi:10.1016/j.amjcard.2005.02.051
- Després JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881-887. doi:10.1038/nature05488
- Liu H, Zhu J, Gao R, et al. Estimating effects of whole grain consumption on type 2 diabetes, colorectal cancer and cardiovascular disease: a burden of proof study. Nutr J. 2024;23(1):49. Published 2024 May 14. doi:10.1186/s12937-024-00957-x
- Alessi MC, Juhan-Vague I. PAI-1 and the metabolic syndrome: links, causes, and consequences. Arterioscler Thromb Vasc Biol. 2006;26(10):2200-2207. doi:10.1161/01.ATV.0000242905.41404.68
- Rytka JM, Wueest S, Schoenle EJ, Konrad D. The portal theory supported by venous drainage-selective fat transplantation. Diabetes. 2011;60(1):56-63. doi:10.2337/db10-0697
- Duell PB, Welty FK, Miller M, et al. Nonalcoholic Fatty Liver Disease and Cardiovascular Risk: A Scientific Statement From the American Heart Association. Arterioscler Thromb Vasc Biol. 2022;42(6):e168-e185. doi:10.1161/ATV.0000000000000153
- Thomas EL, Frost G, Taylor-Robinson SD, Bell JD. Excess body fat in obese and normal-weight subjects. Nutr Res Rev. 2012;25(1):150-161. doi:10.1017/S0954422412000054
- Caleyachetty R, Thomas GN, Toulis KA, et al. Metabolically Healthy Obese and Incident Cardiovascular Disease Events Among 3.5 Million Men and Women. J Am Coll Cardiol. 2017;70(12):1429-1437. doi:10.1016/j.jacc.2017.07.763
- Bello-Chavolla OY, Antonio-Villa NE, Vargas-Vázquez A, et al. Metabolic Score for Visceral Fat (METS-VF), a novel estimator of intra-abdominal fat content and cardio-metabolic health. Clin Nutr. 2020;39(5):1613-1621. doi:10.1016/j.clnu.2019.07.012
- Giannos P, Prokopidis K, Candow DG, et al. Shorter sleep duration is associated with greater visceral fat mass in US adults: Findings from NHANES, 2011-2014. Sleep Med. 2023;105:78-84. doi:10.1016/j.sleep.2023.03.013
- Neeland IJ, Ross R, Després JP, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. 2019;7(9):715-725. doi:10.1016/S2213-8587(19)30084-1
- Bose M, Oliván B, Laferrère B. Stress and obesity: the role of the hypothalamic-pituitary-adrenal axis in metabolic disease. Curr Opin Endocrinol Diabetes Obes. 2009;16(5):340-346. doi:10.1097/MED.0b013e32832fa137
- Kissebah AH, Krakower GR. Regional adiposity and morbidity. Physiol Rev. 1994;74(4):761-811. doi:10.1152/physrev.1994.74.4.761
- Masuzaki H, Paterson J, Shinyama H, et al. A transgenic model of visceral obesity and the metabolic syndrome. Science. 2001;294(5549):2166-2170. doi:10.1126/science.1066285
- Stimson RH, Walker BR. Glucocorticoids and 11beta-hydroxysteroid dehydrogenase type 1 in obesity and the metabolic syndrome. Minerva Endocrinol. 2007;32(3):141-159.
- Masuzaki H, Yamamoto H, Kenyon CJ, et al. Transgenic amplification of glucocorticoid action in adipose tissue causes high blood pressure in mice. J Clin Invest. 2003;112(1):83-90. doi:10.1172/JCI17845
- Chaput JP, Tremblay A. Adequate sleep to improve the treatment of obesity. CMAJ. 2012;184(18):1975-1976. doi:10.1503/cmaj.120876
- Hairston KG, Bryer-Ash M, Norris JM, Haffner S, Bowden DW, Wagenknecht LE. Sleep duration and five-year abdominal fat accumulation in a minority cohort: the IRAS family study. Sleep. 2010;33(3):289-295. doi:10.1093/sleep/33.3.289
- McKeown NM, Troy LM, Jacques PF, Hoffmann U, O’Donnell CJ, Fox CS. Whole- and refined-grain intakes are differentially associated with abdominal visceral and subcutaneous adiposity in healthy adults: the Framingham Heart Study. Am J Clin Nutr. 2010;92(5):1165-1171. doi:10.3945/ajcn.2009.29106
- Monteiro CA, Cannon G, Moubarac JC, Levy RB, Louzada MLC, Jaime PC. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;21(1):5-17. doi:10.1017/S1368980017000234
- Fridén M, Kullberg J, Ahlström H, Lind L, Rosqvist F. Intake of Ultra-Processed Food and Ectopic-, Visceral- and Other Fat Depots: A Cross-Sectional Study. Front Nutr. 2022;9:774718. Published 2022 Apr 4. doi:10.3389/fnut.2022.774718
- Wewege M, van den Berg R, Ward RE, Keech A. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: a systematic review and meta-analysis. Obes Rev. 2017;18(6):635-646. doi:10.1111/obr.12532
- Keating SE, Johnson NA, Mielke GI, Coombes JS. A systematic review and meta-analysis of interval training versus moderate-intensity continuous training on body adiposity. Obes Rev. 2017;18(8):943-964. doi:10.1111/obr.12536
- LaForgia J, Withers RT, Gore CJ. Effects of exercise intensity and duration on the excess post-exercise oxygen consumption. J Sports Sci. 2006;24(12):1247-1264. doi:10.1080/02640410600552064
- Hopkins M, Blundell JE. Energy balance, body composition, sedentariness and appetite regulation: pathways to obesity. Clin Sci (Lond). 2016;130(18):1615-1628. doi:10.1042/CS20160006
- Seidell JC, Bouchard C. Visceral fat in relation to health: is it a major culprit or simply an innocent bystander?. Int J Obes Relat Metab Disord. 1997;21(8):626-631. doi:10.1038/sj.ijo.0800467
- Onitsuka Y, Takeshima F, Ichikawa T, Kohno S, Nakao K. Estimation of visceral fat and fatty liver disease using ultrasound in patients with diabetes. Intern Med. 2014;53(6):545-553. doi:10.2169/internalmedicine.53.1294