Abstract
Atherosclerotic cardiovascular disease (ASCVD) remains the leading global cause of mortality despite major advances in acute care. Modern preventive cardiology increasingly recognizes apolipoprotein B (ApoB)–containing lipoproteins as the necessary and causal drivers of atherosclerosis. Evidence from genetics, epidemiology, and randomized trials supports a cumulative exposure model in which the magnitude and duration of ApoB exposure determine lifetime risk. This review synthesizes foundational biological discoveries, landmark clinical trials, Mendelian randomization studies, and emerging gene-based therapies that collectively define a shift from reactive treatment to primordial prevention.
-
The Modern Prevention Paradigm
Although mortality from acute myocardial infarction has declined substantially, the global burden of ASCVD remains high due to persistent exposure to modifiable risk factors. The PURE study demonstrated that traditional risk factors account for the majority of cardiovascular events across diverse global populations [1].
Preventive cardiology is therefore shifting from treating late-stage events to preventing plaque formation altogether—a strategy consistent with the concept of primordial prevention.
-
LDL Receptors and the Central Role of ApoB
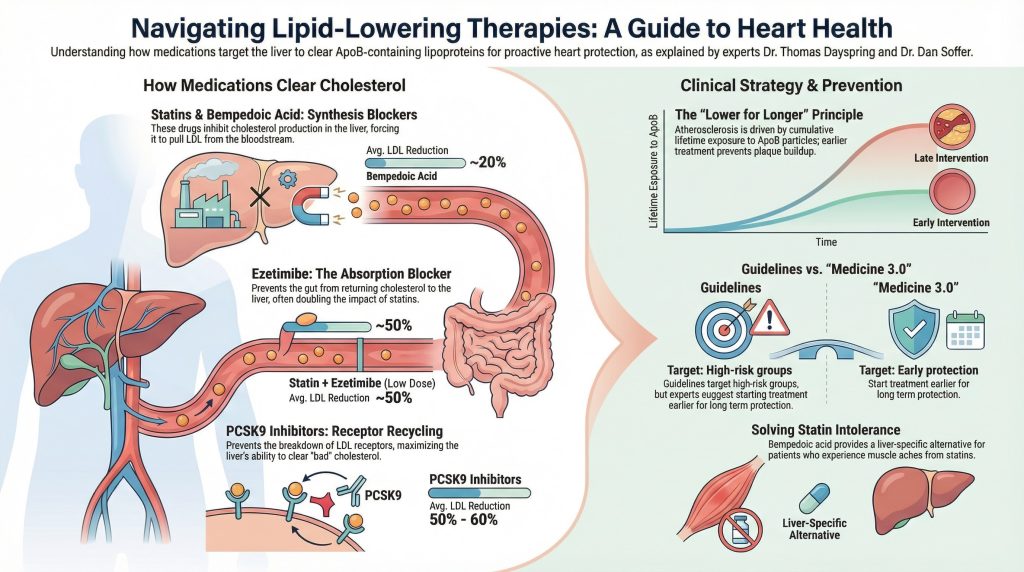
The biological foundation of lipid-lowering therapy rests on the LDL receptor (LDLR) pathway described by Brown and Goldstein [2]. Hepatic LDLR expression increases clearance of circulating ApoB-containing particles (LDL, VLDL remnants, Lp(a)), thereby reducing atherogenic burden.
Statins inhibit HMG-CoA reductase, upregulating LDLR expression. PCSK9 inhibitors prevent LDLR degradation, prolonging receptor recycling. In the FOURIER trial, evolocumab reduced LDL-C by approximately 59% and significantly lowered major cardiovascular events [3]. These results reinforced the principle that greater ApoB reduction yields greater cardiovascular risk reduction.
-
ApoB as the Causal Particle: The Cumulative Exposure Model
The European Atherosclerosis Society consensus statement led by Ference et al. established that ApoB-containing lipoproteins are causal in ASCVD [4]. The total number of circulating atherogenic particles—not LDL-C concentration alone—determines arterial exposure.
Genetic, epidemiologic, and clinical trial data support a cumulative burden model (“cholesterol-years” concept):
Risk ≈ magnitude of ApoB × duration of exposure.
Short-term 10-year risk calculators may underestimate lifetime risk in younger individuals with prolonged exposure ahead of them. The clinical rationale for ApoB measurement as a more accurate representation of atherogenic particle number has been articulated in detail by Sniderman et al. [5].
-
Reassessing HDL: Why Raising HDL Failed
Pharmacologic HDL raising has repeatedly failed to reduce cardiovascular events when ApoB burden remains unchanged.
The AIM-HIGH trial demonstrated no incremental benefit from adding niacin to intensive statin therapy [6]. The HPS2-THRIVE trial similarly showed no meaningful reduction in major vascular events and identified increased adverse events with niacin therapy [7].
These trials shifted focus away from HDL-C as a therapeutic target and reinforced ApoB reduction as the primary modifiable driver of ASCVD risk.
-
Bempedoic Acid: Upstream LDLR Modulation
Bempedoic acid inhibits ATP-citrate lyase (ACL), an upstream enzyme in cholesterol synthesis. It is activated primarily in the liver and not in skeletal muscle, limiting muscle exposure to the active drug.
In the CLEAR Outcomes trial, bempedoic acid reduced major adverse cardiovascular events in statin-intolerant patients [8]. While muscle-related side effects were not significantly increased versus placebo, higher rates of hyperuricemia and gout were observed [8]. Bempedoic acid therefore represents an additional LDLR-mediated strategy for ApoB reduction with a distinct safety profile.
-
Genetics as a Natural Randomized Trial
Mendelian randomization studies provide insight into lifelong LDL/ApoB exposure. Ference et al. demonstrated that lifelong genetically lower LDL-C is associated with substantially greater reductions in coronary heart disease risk than those observed in short-term pharmacologic trials [9].
The magnitude of benefit per 1 mmol/L lower LDL-C from birth exceeded the risk reductions typically observed in 5-year statin trials, supporting the principle that earlier and longer ApoB reduction produces larger lifetime benefit [9].
-
RNA Therapeutics and Gene Editing
PCSK9 siRNA (Inclisiran)
Inclisiran uses small interfering RNA (siRNA) to inhibit hepatic PCSK9 production. In ORION-1 and subsequent phase 3 trials, inclisiran produced durable LDL-C reductions of approximately 50% with maintenance dosing every six months following initial and 3-month doses [10,11].
CRISPR Base Editing (PCSK9)
In primate studies, in vivo CRISPR base editing of PCSK9 resulted in durable LDL-C reductions [12]. This foundational work forms the scientific basis for first-in-human gene-editing trials such as heart-1 (Verve-101), which aim to achieve long-term reduction of ApoB-containing particles through a single intervention [12].
- Comparison of Modern Lipid-Lowering Strategies
| Therapy | Dosing Frequency | Primary Mechanism | Key Evidence |
| Statins | Daily | HMG-CoA reductase inhibition → ↑ LDLR | Landmark statin trials |
| PCSK9 mAbs | Every 2–4 weeks | Prevent LDLR degradation | FOURIER [3] |
| Bempedoic Acid | Daily | ACL inhibition (hepatic activation) | CLEAR Outcomes [8] |
| Inclisiran | Day 1, 3 months, then every 6 months | siRNA blocking PCSK9 production | ORION-1, ORION-10/11 [10,11] |
| PCSK9 Base Editing | Investigational | Permanent gene editing | Primate data [12] |
References
- Yusuf S, Joseph P, Rangarajan S, et al. Modifiable risk factors, cardiovascular disease, and mortality in 155 722 individuals from 21 high-income, middle-income, and low-income countries (PURE): a prospective cohort study. Lancet. 2020;395(10226):795-808. doi:10.1016/S0140-6736(19)32008-2
- Brown MS, Goldstein JL. A receptor-mediated pathway for cholesterol homeostasis. Science. 1986;232(4746):34-47. doi:10.1126/science.3513311
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med. 2017;376(18):1713-1722. doi:10.1056/NEJMoa1615664
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459-2472. doi:10.1093/eurheartj/ehx144
- Sniderman AD, Thanassoulis G, Glavinovic T, et al. Apolipoprotein B Particles and Cardiovascular Disease: A Narrative Review. JAMA Cardiol. 2019;4(12):1287-1295. doi:10.1001/jamacardio.2019.3780
- AIM-HIGH Investigators, Boden WE, Probstfield JL, et al. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365(24):2255-2267. doi:10.1056/NEJMoa1107579
- HPS2-THRIVE Collaborative Group, Landray MJ, Haynes R, et al. Effects of extended-release niacin with laropiprant in high-risk patients. N Engl J Med. 2014;371(3):203-212. doi:10.1056/NEJMoa1300955
- Nissen SE, Lincoff AM, Brennan D, et al. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients. N Engl J Med. 2023;388(15):1353-1364. doi:10.1056/NEJMoa2215024
- Ference BA, Yoo W, Alesh I, et al. Effect of long-term exposure to lower low-density lipoprotein cholesterol beginning early in life on the risk of coronary heart disease: a Mendelian randomization analysis. J Am Coll Cardiol. 2012;60(25):2631-2639. doi:10.1016/j.jacc.2012.09.017
- Ray KK, Landmesser U, Leiter LA, et al. Inclisiran in Patients at High Cardiovascular Risk with Elevated LDL Cholesterol. N Engl J Med. 2017;376(15):1430-1440. doi:10.1056/NEJMoa1615758
- Ray KK, Wright RS, Kallend D, et al. Two Phase 3 Trials of Inclisiran in Patients with Elevated LDL Cholesterol. N Engl J Med. 2020;382(16):1507-1519. doi:10.1056/NEJMoa1912387
- Musunuru K, Chadwick AC, Mizoguchi T, et al. In vivo CRISPR base editing of PCSK9 durably lowers cholesterol in primates. Nature. 2021;593(7859):429-434. doi:10.1038/s41586-021-03534-y