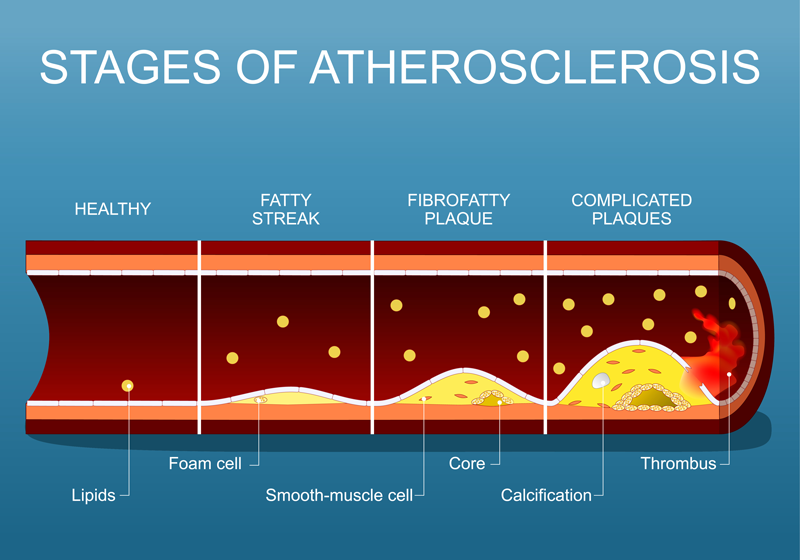
How we think about cardiovascular risk has changed a lot. We used to focus on total cholesterol and later LDL cholesterol (LDL-C) as the main villains. Now, the picture is more precise: risk is driven by how many atherogenic particles are circulating, which genetically “high-risk” particles are present, and how much inflammation is active in the vessel wall. At the heart of this newer view is a practical triad of biomarkers: apolipoprotein B-100 (ApoB), which counts the number of atherogenic lipoprotein particles;¹ lipoprotein(a) [Lp(a)], a mostly inherited LDL-like particle with added thrombotic risk;² and high-sensitivity C-reactive protein (hsCRP), a marker that tracks systemic and vascular inflammation.³
LDL-C still matters and remains the standard therapeutic target, but clinical experience (and growing evidence) shows that LDL-C can miss important risk—especially when ApoB is high, Lp(a) is elevated, or hsCRP suggests ongoing inflammation.⁴ In many patients, these three factors stack together and create risk that feels “out of proportion” to traditional lipid panels. When you compare this biochemical and genetic risk profile with behavioral insults like cigarette smoking, the hierarchy of risk becomes even more nuanced, reinforcing the need for personalized prevention strategies in both primary and secondary prevention.⁵
The Molecular Framework of Apolipoprotein B-100 and Particle Pathogenicity
Atherosclerosis doesn’t start because the blood contains “too much cholesterol” in the abstract. It starts because too many atherogenic particles are circulating, and a fraction of them get trapped in the artery wall.⁶ The single best way to understand this is to think in terms of particle number, not just cholesterol mass.⁶
Apolipoprotein B (ApoB) is the key structural protein on all potentially atherogenic lipoproteins: very-low-density lipoproteins (VLDL), intermediate-density lipoproteins (IDL), low-density lipoproteins (LDL), and also lipoprotein(a).⁸ Each of these particles carries exactly one ApoB-100 molecule, which makes ApoB a convenient biological “counter”: the ApoB concentration in plasma tells you how many atherogenic particles are present.⁷
This is why relying exclusively on LDL-C can be misleading in some common clinical settings—especially hypertriglyceridemia, metabolic syndrome, and type 2 diabetes.⁶ In these states, LDL particles often carry less cholesterol per particle, shifting toward small, dense LDL (sdLDL). That means a patient can show an “acceptable” LDL-C value while still having a high number of LDL particles—a pattern often described as LDL-C/ApoB discordance.⁹ ApoB measurement bypasses that limitation by directly reflecting particle burden, which better captures the likelihood of lipoprotein entry into the arterial intima.⁷
Mechanisms of Subendothelial Infiltration and Retention
Atherosclerosis begins when the endothelium—normally a smooth barrier—becomes more permeable or dysfunctional, often due to shear stress, oxidative injury, metabolic dysfunction, or chemical exposure (including tobacco smoke).¹¹ Once that barrier is compromised, ApoB-containing particles can move into the arterial intima.¹⁰
What matters next is not only entry, but retention. In the subendothelial space, ApoB particles interact with the extracellular matrix rather than simply drifting by concentration gradients.¹² ApoB-100 contains positively charged regions that bind to negatively charged sulfate groups on arterial proteoglycans. This electrostatic interaction is one of the reasons particles become “stuck” in the vessel wall—an essential early step in plaque formation.¹⁰ Once trapped, particles undergo oxidative and enzymatic modification, generating oxidized lipoproteins that are far more inflammatory and immunogenic than native particles.¹³
These modified particles act like danger signals. They recruit monocytes, promote macrophage uptake through scavenger receptors, and drive the formation of lipid-laden foam cells—one of the earliest histologic hallmarks of atherosclerotic lesions.¹³
Stoichiometry and Diagnostic Precision of ApoB
ApoB’s clinical advantage is not only conceptual—it’s practical. Much of routine LDL-C reporting still depends on calculated methods, most commonly the Friedewald equation:¹⁴
This approach becomes less accurate when triglycerides are elevated (typically when TG exceed ~3.5–4 mmol/L) or when blood is drawn in a non-fasting state.¹⁴ ApoB, in contrast, is measured directly through immunoassays (immunoturbidimetric or immunonephelometric methods) that have been internationally standardized.¹⁵ In practice, ApoB tends to show lower analytic bias and better reproducibility than calculated lipid measures, which is why it is increasingly favored for assessing particle-driven risk.⁷
| Physiological Metric | Diagnostic Method | Sensitivity to Fasting |
| LDL-C | Cholesterol mass | Calculation (Friedewald) |
| ApoB-100 | Particle count (1:1 ratio) | Direct measurement |
| Non-HDL-C | All atherogenic cholesterol | Calculation (TC − HDL-C) |
| Lp(a) | Genetic particle subtype | Immunoturbidimetric |
Lipoprotein(a): The Genetic Vanguard of Atherothrombosis
Lp(a) is one of the most clinically important (and frustrating) lipoproteins because it is largely genetically determined and minimally affected by lifestyle changes. Structurally, Lp(a) looks like an LDL particle with an attached additional protein—apolipoprotein(a) [apo(a)]—linked to ApoB-100 by a disulfide bond.² It’s this additional apo(a) component that makes Lp(a) biologically distinctive and often more dangerous than standard LDL. Some estimates suggest it may be several-fold more potent as a driver of cardiovascular disease than LDL alone.¹⁶
Genetic Regulation and Kringle IV Complexity
Lp(a) levels are mainly controlled by the LPA gene on chromosome 6q26–q27. Lp(a) concentration is typically 70% to 90% heritable and remains relatively stable over a lifetime, unlike LDL-C which can shift substantially with diet, weight loss, and medications.¹⁷ The striking variability in Lp(a) between individuals—sometimes over a 1000-fold range—comes largely from copy-number variation in the Kringle IV type 2 (KIV2) repeats within apo(a).¹⁸
Kringle domains are looped structures stabilized by disulfide bonds. Apo(a) contains multiple kringle subtypes (KIV1–KIV10), but KIV2 is the one that varies widely across people. Those with fewer KIV2 repeats generally make smaller apo(a) isoforms and tend to have higher plasma Lp(a) levels. This inverse relationship between apo(a) size and Lp(a) concentration explains much of the genetic contribution to cardiovascular risk from Lp(a).¹⁹
The Dual Mechanisms of Lp(a) Pathogenicity
Lp(a) increases risk through two main pathways that overlap in real-world disease: a pro-atherogenic/pro-inflammatory pathway and a pro-thrombotic/anti-fibrinolytic pathway.²²
Atherogenic and pro-inflammatory drive: Like other ApoB particles, Lp(a) can cross the endothelium and accumulate in the intima. But Lp(a) is also a major carrier of oxidized phospholipids (OxPL) in plasma.²⁰ OxPL behave like strong inflammatory ligands, promoting endothelial activation, smooth muscle proliferation, macrophage dysfunction, and sometimes apoptosis—features that contribute to plaque growth and instability.²⁰
Thrombotic and anti-fibrinolytic interference: Apo(a) shares significant structural homology with plasminogen.²¹ Because of this resemblance, Lp(a) can compete with plasminogen for binding sites on fibrin, interfering with plasmin generation and impairing fibrinolysis.²² In effect, Lp(a) encourages thrombus persistence, increasing the chance that plaque rupture leads to a clinically significant occlusive event such as myocardial infarction.²²
Systemic Inflammation and the Sentinel Role of hsCRP
Atherosclerosis is now widely understood as a chronic inflammatory condition affecting the arterial wall.²³ Among the inflammatory biomarkers available clinically, hsCRP remains the most commonly used and best standardized.³ hsCRP does not tell you where inflammation is coming from, but persistent low-grade elevation strongly correlates with vascular inflammatory risk.³
The NLRP3 Inflammasome and CRP Induction
When cholesterol crystals and oxidized ApoB particles build up in the intima, they activate immune pathways, including the NLRP3 inflammasome in macrophages.²⁴ This leads to processing of pro-interleukin-1β and pro-IL-18 into active cytokines.²⁴ These cytokines stimulate downstream IL-6 signaling, which triggers the liver to synthesize and release CRP.²⁵
CRP can spike dramatically during infection, but chronically elevated hsCRP (often defined as hsCRP ≥ 2 mg/L) behaves more like a “smoke detector” for ongoing vascular inflammation and future cardiovascular events.³
Lessons from the JUPITER Trial
The JUPITER trial was a turning point because it showed that inflammatory risk can identify high-risk patients even when LDL-C looks fine.²⁶ The study enrolled individuals with LDL-C below usual treatment thresholds (<130 mg/dL) but with hsCRP ≥2.0 mg/L. Participants receiving rosuvastatin 20 mg daily had a 44% reduction in major cardiovascular events.²⁶ Clinically, the takeaway was simple: some patients carry substantial risk through inflammation even when they do not appear “hyperlipidemic” by LDL-C alone.
Mapping the Interplay: Synergistic Risk and Pathogenic Cross-talk
ApoB, Lp(a), and hsCRP do not operate in isolation. Their relationship is better described as interactive, with overlapping mechanisms that can amplify each other’s harm.⁴
High ApoB means more particles enter the vessel wall and more substrate becomes available for oxidative modification. Those modified particles intensify inflammation, raising hsCRP. Inflammation then further disrupts endothelial function, making it easier for additional ApoB particles to enter—creating a self-reinforcing loop.²³
This synergy shows up clearly in large population studies. In a study of over 320,000 UK Biobank participants, LDL-C, Lp(a), and hsCRP were each independently associated with major adverse cardiovascular events (MACE), but the combined effect was far greater than any single marker alone.⁴
| Biomarker Risk Strata | MACE Risk Elevation (Non-users of Statins) |
| All Markers Low | 1.00 (Reference) |
| High LDL-C Only | +13% risk per SD |
| High Lp(a) Only | +8% risk per SD |
| High hsCRP Only | +6% risk per SD |
| Triple Elevation | +77% risk (HR 1.77) |
Comparison of Biomarker Risks to the Pathogenic Impact of Smoking
Smoking remains one of the most aggressive cardiovascular toxins because it generates oxidative stress, drives chronic inflammation, and directly injures the endothelium.²⁷ The question clinicians often ask is: how does smoking compare to lipid and biomarker risk?
INTERHEART provides one of the most useful comparisons because it included diverse populations across 52 countries.⁵ It showed that current smoking had one of the highest individual odds ratios for MI, but dyslipidemia (measured by ApoB/ApoA1 ratio) carried an even larger population attributable risk, because dyslipidemia is so common globally.²⁸
| Risk Factor | Odds Ratio (OR) for MI | Population Attributable Risk (PAR) |
| Current Smoking | 3.63 | 35.7% (Global) |
| High ApoB/ApoA1 Ratio | 3.43 | 54.1% (Global) |
| Diabetes Mellitus | 3.42 | 16.4% |
| Hypertension | 1.89 | 10.7% |
The Female Paradox: Sex-Specific Risk Sensitivities
Large cohorts show that women may experience a greater relative increase in MI risk from certain exposures, especially smoking and metabolic dysfunction.²⁹ In UK Biobank, current smoking was linked to a hazard ratio for MI of 3.46 in women compared with 2.23 in men, producing a ratio of hazard ratios (RHR) of 1.55.²⁹
Clinical Implications: Managing the Residual Risk Triad
Even with excellent statin therapy and strong LDL-C lowering, cardiovascular events still occur. This is often referred to as residual risk, and it commonly reflects a combination of residual particle risk (ApoB), genetic risk (Lp(a)), and residual inflammatory risk (hsCRP).³⁰
Statins lower LDL-C and reduce events, but they have little effect on Lp(a), and multiple studies suggest statins may increase Lp(a) modestly (often ~10–20%).³¹ PCSK9 inhibitors reduce LDL-C substantially and also lower Lp(a) by about ~27%.³²
The most promising future approach is direct Lp(a) lowering using antisense oligonucleotides (ASOs) or siRNA platforms, which have shown 80–90% reductions in early studies.³³ Finally, inflammation-focused trials such as CANTOS demonstrated that reducing inflammatory signaling (independent of lipids) can reduce MACE, reinforcing the clinical reality that inflammation is not just a bystander.³⁴
Conclusion: The Integrated Risk Map
Preventive cardiology is increasingly moving from a single-marker “cholesterol hypothesis” toward a more integrated approach. In practical terms, ApoB tells you particle burden, Lp(a) tells you inherited atherothrombotic risk, and hsCRP tells you about inflammatory activation. When these risks cluster, events can occur despite “good” LDL-C numbers.
Used together, these markers support more individualized decisions about therapy intensity and emerging targeted treatments—aimed at achieving the deepest possible reduction in cardiovascular risk.
References
- Nicholls SJ, Nelson AJ. Current perspectives on Lp(a)-lowering therapies: Who may benefit?. Kardiol Pol. 2025;83(6):688-694. doi:10.33963/v.phj.106327
- Tsimikas S. A Test in Context: Lipoprotein(a): Diagnosis, Prognosis, Controversies, and Emerging Therapies. J Am Coll Cardiol. 2017;69(6):692-711. doi:10.1016/j.jacc.2016.11.042
- Ridker PM. High-sensitivity C-reactive protein: potential adjunct for global risk assessment in the primary prevention of cardiovascular disease. Circulation. 2001;103(13):1813-1818. doi:10.1161/01.cir.103.13.1813
- Markus MRP, Ittermann T, Mariño Coronado J, et al. Low-density lipoprotein cholesterol, lipoprotein(a) and high-sensitivity C-reactive protein are independent predictors of cardiovascular events. Eur Heart J. 2025;46(39):3863-3874. doi:10.1093/eurheartj/ehaf281
- Yusuf S, Hawken S, Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):937-952. doi:10.1016/S0140-6736(04)17018-9
- Sniderman AD, Navar AM, Thanassoulis G. Apolipoprotein B vs Low-Density Lipoprotein Cholesterol and Non-High-Density Lipoprotein Cholesterol as the Primary Measure of Apolipoprotein B Lipoprotein-Related Risk: The Debate Is Over. JAMA Cardiol. 2022;7(3):257-258. doi:10.1001/jamacardio.2021.5080
- Contois JH, McConnell JP, Sethi AA, et al. Apolipoprotein B and cardiovascular disease risk: position statement from the AACC Lipoproteins and Vascular Diseases Division Working Group on Best Practices. Clin Chem. 2009;55(3):407-419. doi:10.1373/clinchem.2008.118356
- Pearson GJ, Thanassoulis G, Anderson TJ, et al. 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults. Can J Cardiol. 2021;37(8):1129-1150. doi:10.1016/j.cjca.2021.03.016
- Otvos JD, Mora S, Shalaurova I, Greenland P, Mackey RH, Goff DC Jr. Clinical implications of discordance between low-density lipoprotein cholesterol and particle number. J Clin Lipidol. 2011;5(2):105-113. doi:10.1016/j.jacl.2011.02.001
- Tabas I, Williams KJ, Borén J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation. 2007;116(16):1832-1844. doi:10.1161/CIRCULATIONAHA.106.676890
- Messner B, Bernhard D. Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol. 2014;34(3):509-515. doi:10.1161/ATVBAHA.113.300156
- Borén J, Williams KJ. The central role of arterial retention of cholesterol-rich apolipoprotein-B-containing lipoproteins in the pathogenesis of atherosclerosis: a triumph of simplicity. Curr Opin Lipidol. 2016;27(5):473-483. doi:10.1097/MOL.0000000000000330
- Miller YI, Choi SH, Wiesner P, et al. Oxidation-specific epitopes are danger-associated molecular patterns recognized by pattern recognition receptors of innate immunity. Circ Res. 2011;108(2):235-248. doi:10.1161/CIRCRESAHA.110.223875
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499-502.
- Albers JJ, Marcovina SM. Standardization of apolipoprotein B and A-I measurements. Clin Chem. 1989;35(7):1357-1361.
- Kamstrup PR, Benn M, Tybjaerg-Hansen A, Nordestgaard BG. Extreme lipoprotein(a) levels and risk of myocardial infarction in the general population: the Copenhagen City Heart Study. Circulation. 2008;117(2):176-184. doi:10.1161/CIRCULATIONAHA.107.715698
- Boerwinkle E, Leffert CC, Lin J, Lackner C, Chiesa G, Hobbs HH. Apolipoprotein(a) gene accounts for greater than 90% of the variation in plasma lipoprotein(a) concentrations. J Clin Invest. 1992;90(1):52-60. doi:10.1172/JCI115855
- Sandholzer C, Hallman DM, Saha N, et al. Effects of the apolipoprotein(a) size polymorphism on the lipoprotein(a) concentration in 7 ethnic groups. Hum Genet. 1991;86(6):607-614. doi:10.1007/BF00201550
- Koschinsky ML, Marcovina SM. Structure-function relationships in apolipoprotein(a): insights into lipoprotein(a) assembly and pathogenicity. Curr Opin Lipidol. 2004;15(2):167-174. doi:10.1097/00041433-200404000-00009
- Bergmark C, Dewan A, Orsoni A, et al. A novel function of lipoprotein [a] as a preferential carrier of oxidized phospholipids in human plasma. J Lipid Res. 2008;49(10):2230-2239. doi:10.1194/jlr.M800174-JLR200
- McLean JW, Tomlinson JE, Kuang WJ, et al. cDNA sequence of human apolipoprotein(a) is homologous to plasminogen. Nature. 1987;330(6144):132-137. doi:10.1038/330132a0
- Boffa MB, Marcovina SM, Koschinsky ML. Lipoprotein(a) as a risk factor for atherosclerosis and thrombosis: mechanistic insights from animal models. Clin Biochem. 2004;37(5):333-343. doi:10.1016/j.clinbiochem.2003.12.007
- Libby P. Inflammation in atherosclerosis. Nature. 2002;420(6917):868-874. doi:10.1038/nature01323
- Duewell P, Kono H, Rayner KJ, et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464(7293):1357-1361. doi:10.1038/nature08938
- Ridker PM. From C-Reactive Protein to Interleukin-6 to Interleukin-1: Moving Upstream To Identify Novel Targets for Atheroprotection. Circ Res. 2016;118(1):145-156. doi:10.1161/CIRCRESAHA.115.306656
- Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359(21):2195-2207. doi:10.1056/NEJMoa0807646
- Ambrose JA, Barua RS. The pathophysiology of cigarette smoking and cardiovascular disease: an update. J Am Coll Cardiol. 2004;43(10):1731-1737. doi:10.1016/j.jacc.2003.12.047
- McQueen MJ, Hawken S, Wang X, et al. Lipids, lipoproteins, and apolipoproteins as risk markers of myocardial infarction in 52 countries (the INTERHEART study): a case-control study. Lancet. 2008;372(9634):224-233. doi:10.1016/S0140-6736(08)61076-4
- Millett ERC, Peters SAE, Woodward M. Sex differences in risk factors for myocardial infarction: cohort study of UK Biobank participants. BMJ. 2018;363:k4247. Published 2018 Nov 7. doi:10.1136/bmj.k4247
- Ridker PM. Residual inflammatory risk: addressing the obverse side of the atherosclerosis prevention coin. Eur Heart J. 2016;37(22):1720-1722. doi:10.1093/eurheartj/ehw024
- Tsimikas S, Gordts PLSM, Nora C, Yeang C, Witztum JL. Statin therapy increases lipoprotein(a) levels. Eur Heart J. 2020;41(24):2275-2284. doi:10.1093/eurheartj/ehz310
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med. 2017;376(18):1713-1722. doi:10.1056/NEJMoa1615664
- Tsimikas S, Karwatowska-Prokopczuk E, Gouni-Berthold I, et al. Lipoprotein(a) Reduction in Persons with Cardiovascular Disease. N Engl J Med. 2020;382(3):244-255. doi:10.1056/NEJMoa1905239
- Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N Engl J Med. 2017;377(12):1119-1131. doi:10.1056/NEJMoa1707914