Most people think there’s one perfect diet for a long life. But emerging science suggests something far more interesting: the rules of healthy eating actually change as we age. What protects you at 45 may not protect you at 85. Longevity isn’t about sticking to the same nutrition plan forever — it’s about adapting to your body’s shifting biology. And according to new research, age 65 appears to be a major turning point.
Before 65, the body tends to benefit from keeping growth signals under control. Eating moderately — especially not overdoing protein and calories — may help lower growth hormones that are linked to cancer and other age-related diseases. This stage of life is about protection and maintenance. Giving your body time to repair itself through balanced eating, and possibly time-restricted eating, supports important cellular cleanup processes that help preserve long-term health.
After 65, however, the priorities shift dramatically. The biggest threats are no longer rapid growth diseases, but muscle loss, frailty, falls, and loss of independence. At this stage, protein becomes critically important. Research in adults over 85 shows that higher protein intake is associated with lower mortality and better functional independence. In simple terms, earlier in life you focus on not overstimulating growth — later in life, you focus on preserving strength.
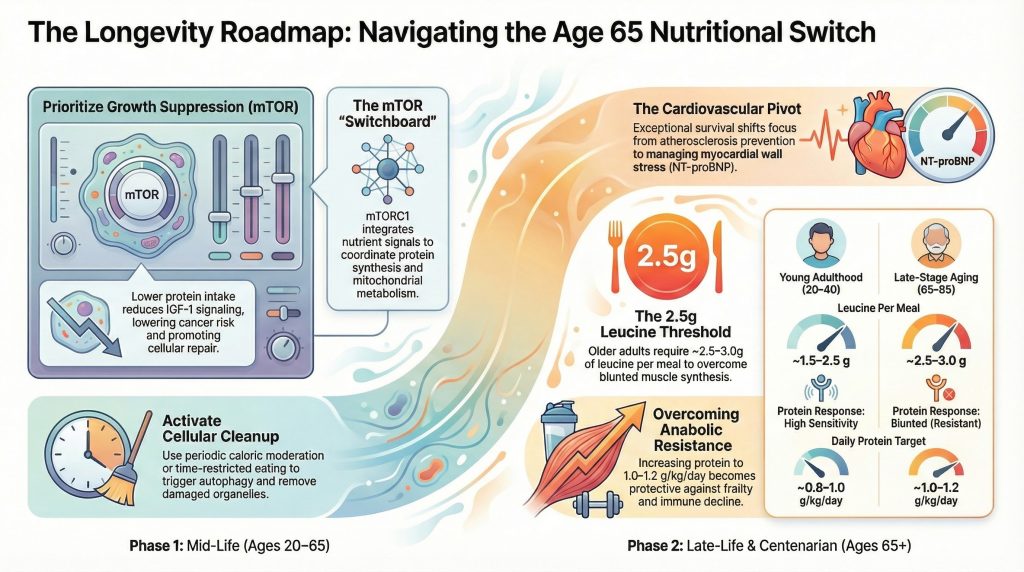
One reason for this shift lies in a biological system called mTOR, a master switch inside your cells that decides whether they grow or repair themselves. When constantly activated by high nutrient intake, cells stay in growth mode and don’t clean up internal damage efficiently. But as we age, the issue isn’t excessive growth — it’s maintaining muscle and resilience. That’s where adequate protein intake becomes protective rather than harmful.
Aging muscles also become less responsive to protein, a phenomenon known as anabolic resistance. In practical terms, older adults need more protein per meal to trigger muscle maintenance. An amino acid called leucine acts like the ignition key for muscle building. After 65, aiming for roughly 25–40 grams of high-quality protein per meal — enough to provide about 2.5 to 3 grams of leucine — can help overcome this resistance. Standard dietary recommendations often fall short for older adults.
Heart health also plays a powerful role in longevity. The condition of your arteries directly affects your brain. Stiff or inflamed blood vessels increase the risk of cognitive decline and dementia. Studies of people who live past 100 show they tend to maintain lower levels of heart stress markers, suggesting that protecting cardiovascular health may be one of the most important strategies for preserving both heart and brain function.
Another hallmark of exceptional longevity is efficient cellular “housekeeping.” Centenarians appear to be better at clearing out damaged mitochondria — the tiny power plants inside cells. When mitochondria function well, the body can efficiently use fuel and maintain metabolic flexibility. When they become damaged and accumulate, aging accelerates. Regular exercise, metabolic health, and avoiding chronic overeating remain some of the most reliable ways to support this cleanup system.
Taken together, the message is clear: longevity requires a strategic pivot. From ages 20 to 65, the focus should be on metabolic balance, avoiding excess, and supporting cellular repair. From 65 onward, the emphasis shifts toward preserving muscle, strength, and independence through higher protein intake and resistance training. Beyond 80, maintaining adequate nutrition and cardiovascular stability becomes the central goal.
Living to 100 isn’t about following one rigid diet for decades. It’s about recognizing that your body at 70 has different needs than your body at 40 — and adjusting accordingly. The real key to longevity may not be eating less or eating more, but knowing when to change.
DEEP DIVE HERE: READ MORE BELOW
The mTOR Pathway: A Master Regulatory Hub in Aging and Lifespan
mTOR functions as a conserved metabolic “switchboard” integrating cellular energy status, growth factors, and amino acid availability to coordinate protein synthesis, autophagy, mitochondrial metabolism, and stress responses [1,2]. Across model organisms—including yeast, flies, and mice—age-associated increases in anabolic signaling and reduced autophagic clearance are repeatedly linked to functional decline and shortened lifespan, implicating excessive mTORC1 activity as a contributor to aging biology rather than a neutral correlate [1–3].
Molecular Architecture of mTOR Complexes
mTOR exists in two functionally and structurally distinct complexes: mTORC1 and mTORC2. While both share the catalytic mTOR subunit, their scaffolds determine substrate specificity and biological outputs. mTORC1 is the nutrient-sensitive complex, defined by RAPTOR, and regulates translation through targets such as S6K1 and 4E-BP1 [2,3]. mTORC2 is defined by RICTOR and plays a major role in cytoskeletal organization and Akt-mediated survival signaling [2].
Hyperactivation of mTORC1 in mid- and late life is mechanistically linked to age-associated pathology, including cancer-promoting growth signaling, impaired proteostasis, metabolic dysfunction, and vascular disease [2–4]. By promoting continuous translation while suppressing autophagy (including mitophagy), excessive mTORC1 activity can accelerate accumulation of damaged organelles and misfolded proteins—functionally “clogging” cellular maintenance systems and amplifying senescence-associated phenotypes [2,4].
Table 1. Structural Components and Regulatory Functions of mTORC1
| Component | Function | Role in aging signaling |
| mTOR | Catalytic Ser/Thr kinase | Master integrator of nutrient and energy signals [2] |
| RAPTOR | Substrate recruitment scaffold | Enables phosphorylation of S6K1 and 4E-BP1 [2,3] |
| mLST8 | Kinase-domain stabilizer | Supports structural integrity and signaling output [2] |
| DEPTOR | Endogenous inhibitor | Reduced expression/activity can disinhibit mTOR signaling [2] |
| PRAS40 | Akt-regulated inhibitor | Links insulin/Akt signaling to mTORC1 activation [2] |
| Tti1/Tel2 | Assembly/stability factors | Required for proper complex formation [2] |
Interventions targeting mTOR. Pharmacologic inhibition of mTORC1 with rapamycin and nutritional interventions such as caloric restriction (CR) are among the most reproducible lifespan- and healthspan-extending strategies across model systems [1–3]. In mice, rapamycin can extend lifespan even when initiated later in life, supporting the view that mTOR remains modifiable across the aging trajectory [1]. CR consistently downshifts growth signaling, enhances autophagy, and preserves metabolic efficiency. It is more accurate to describe mTOR inhibition/CR as among the most consistent pro-longevity interventions across taxa rather than the only such intervention, as multiple manipulations (eg, genetic insulin/IGF signaling changes) also extend lifespan in specific organisms [1–3].
The Anabolic Resistance Continuum and Leucine Signaling
As humans transition from young adulthood to late-stage aging (often operationalized as ≥65 years), skeletal muscle progressively exhibits anabolic resistance—a blunted stimulation of muscle protein synthesis (MPS) in response to dietary protein and resistance exercise [5,6]. In younger individuals, modest high-quality protein doses can trigger robust MPS. In older adults, the “anabolic threshold” rises; greater essential amino acid (EAA) availability—particularly leucine—is needed to activate translation initiation and achieve comparable MPS responses [5–7]. Contributing mechanisms include impaired perfusion and amino acid delivery, reduced insulin sensitivity, altered muscle intracellular signaling, and chronic low-grade inflammation [5,6].
The Sestrin2–Leucine Sensor Mechanism
Leucine is distinctive among branched-chain amino acids because it acts as both substrate and signal for mTORC1 activation. Sestrin2 functions as a cytosolic leucine sensor that regulates mTORC1 through the GATOR2 complex and Rag GTPases [8,9]. In low-leucine conditions, Sestrin2 restrains GATOR2, suppressing Rag activation and limiting mTORC1 lysosomal recruitment [8,9]. When leucine is abundant, it binds a conserved hydrophobic pocket on Sestrin2, triggering a conformational shift that disrupts Sestrin2–GATOR2 interaction and permits downstream activation of mTORC1 [8,9]. Structural work demonstrates specificity determinants that favor leucine over valine/isoleucine, including hydrophobic complementarity and coordinated recognition of leucine’s amino and carboxyl groups [8,9].
Thresholds for Bypassing Anabolic Resistance
For practical geronutrition, the key issue is not whether leucine signals mTORC1, but how much leucine exposure is needed to overcome anabolic resistance. Observational breakpoint analyses in older adults have identified a plateau for daily leucine intake associated with lower-limb muscle mass and strength at approximately 7.6–8.0 g/day, equivalent to roughly 110–115 mg/kg/day in typical older cohorts [10]. In parallel, controlled feeding and expert consensus guidance commonly emphasize that older adults often require ~2.5–3.0 g leucine per meal (usually delivered by ~25–40 g high-quality protein) to robustly stimulate MPS [6,7]. Indicator amino acid oxidation methods further suggest leucine requirements in older adults may exceed current recommendations, supporting the concept that leucine can become limiting when protein quality or total intake is low [11].
Table 2. Comparative Protein and Leucine Targets Across the Aging Spectrum
| Metric | Young adulthood (20–40) | Late-stage aging (65–85) | Centenarians (100+) |
| Protein RDA (minimum) | 0.8 g/kg/day | 0.8 g/kg/day (often insufficient) | 0.8 g/kg/day (often insufficient) |
| Practical daily protein target | ~0.8–1.0 g/kg/day | ~1.0–1.2 g/kg/day (higher if frail) [6] | individualized; often ≥1.0 g/kg/day if tolerated and safe [6] |
| Per-meal protein | ~20–30 g | ~25–40 g (distributed evenly) [6,7] | similar or higher density per meal if appetite limited |
| Leucine per meal | ~1.5–2.5 g | ~2.5–3.0 g (leucine-dense) [6,7] | high density often required; appetite constraints dominate |
| Anabolic response | high sensitivity | blunted (resistant) [5] | severe resistance likely; distribution critical |
| Autophagy status | relatively efficient | often impaired | heterogeneous; may be selectively preserved in resilient phenotypes |
The clinical implication is that strict reliance on the standard protein RDA may be inadequate for preserving mobility and independence in many older adults, particularly when appetite declines and energy intake falls [6]. Protein quality, leucine density, and distribution across meals become increasingly central to maintaining functional reserve.
Cardiovascular Transitions: From Atherosclerosis to Heart Failure and Dementia
In the context of longevity, the cardiovascular system follows a distinct trajectory of adaptation and decline. Subclinical atherosclerosis is common in the 70s and 80s, but among the oldest old, dominant causes of disability and death often shift toward heart failure, frailty, and dementia, reflecting cumulative myocardial remodeling, arterial stiffening, and microvascular dysfunction [12–14]. This does not imply coronary disease disappears; rather, the clinical expression of vascular aging increasingly involves brain and heart failure phenotypes.
Subclinical Atherosclerosis and Cognitive Risk
Coronary artery calcification (CAC) is a robust marker of cumulative atherosclerotic burden. Multiple studies link higher CAC to worse brain structure and function, as well as increased risk for cognitive impairment and dementia endpoints in older adults [12,13]. Recent work also suggests that progression of CAC may carry incremental dementia risk signals in prospective cohorts [14]. The mechanistic bridge is plausibly shared vascular aging: endothelial dysfunction, impaired cerebral perfusion, microinfarction burden, and pro-inflammatory signaling that accelerates neurodegenerative vulnerability [15].
Vascular Stiffness as a Driver of Endothelial Senescence
A critical concept in cardiovascular aging is that vascular stiffness is not merely the consequence of plaque accumulation, but can function as a mechanobiological driver of endothelial dysfunction. Stiffening driven by elastin degradation, collagen remodeling, and cross-linking increases endothelial stress and alters mechanotransduction pathways. Experimental and integrative vascular-aging literature supports that increased stiffness and altered flow promote pro-senescent signaling programs and inflammatory mediator production consistent with endothelial aging phenotypes [16,17]. This contributes to a feed-forward loop in which stiffness amplifies inflammation and remodeling, and inflammation further accelerates extracellular matrix dysfunction [16,17].
Table 3. Cardiovascular Biomarkers and Risk Context in Exceptional Longevity
| Biomarker / feature | Standard aging (80s) | Centenarians (100+) | Implications for survival |
| NT-proBNP | often elevated with age | relatively lower in exceptionally surviving subgroups | lower NT-proBNP associated with survival advantage at highest ages [18] |
| CAC burden | commonly present | variable; lower burden supports brain/vascular resilience | higher CAC linked to worse brain outcomes and dementia risk [12–14] |
| Vascular stiffness | often increased | preserved elasticity in resilient phenotypes | reduces endothelial stress and supports microvascular health [16,17] |
| Albumin | declines with age/inflammation | higher albumin signals better reserve | low albumin associates with higher mortality across age groups [18] |
A particularly strong and reproducible marker in studies of the “oldest old” is NT-proBNP. In aggregated longitudinal cohorts including centenarians and (semi-)supercentenarians, the relationship between NT-proBNP and mortality remains robust even after accounting for traditional risk factors and measures of organ reserve [18]. These findings support the view that resistance to myocardial wall stress and subclinical heart failure physiology is a key component of exceptional survival.
The Nutritional “Switch”: The Critical Threshold Around Age 65
One of the most important translational insights in longevity research is that the optimal nutritional strategy is not static across the lifespan. A major “metabolic switch” occurs around ~65 years, where the risk–benefit ratio associated with protein intake and growth signaling changes meaningfully.
The Middle-Age Low-Protein Benefit
During midlife, lower protein intake patterns have been associated with lower IGF-1 signaling and reduced cancer risk in some cohorts, consistent with the view that chronic growth signaling can be deleterious when repair and surveillance mechanisms begin to degrade [19]. In this phase, the nutritional goal often emphasizes insulin sensitivity, metabolic health, and periodic activation of maintenance pathways such as autophagy—achieved through patterns like modest protein intake, time-restricted eating, or caloric moderation [2,19].
The Late-Life High-Protein Requirement
After ~65, the risk profile flips. Low protein intake becomes strongly linked to adverse outcomes driven by sarcopenia, frailty, falls, and reduced immune competence [6,19]. Higher protein intake supports maintenance of muscle mass, functional reserve, and immune responsiveness—systems that become increasingly vulnerable with age [6]. In older adults, protein distribution and leucine density become particularly important as appetite declines and energy needs fall, making nutrient density (not just calories) decisive.
Consistent with this, a prospective study in Japanese adults aged 85–89 years without baseline disability (Kawasaki Aging and Wellbeing Project) reported lower all-cause mortality risk in the highest versus lowest protein intake groups after adjustment for clinical covariates, supporting the concept that higher protein intake can be protective in very old age when functional independence is maintained [20]. Importantly, protein source and food context remain relevant; broader cohort evidence suggests that substituting plant proteins for some animal protein sources—particularly red/processed meat—may improve long-term outcomes in general populations [21].
Proteomic and Genetic Markers of Centenarian Resilience
Centenarians do not “escape” aging; they often delay disease onset and compress morbidity. Systems-level profiling increasingly supports the existence of protective biomarker constellations in extreme longevity cohorts. Large-scale biomarker work in the oldest old identifies profiles consistent with preserved organ reserve and lower heart-failure physiology among those reaching the highest ages, with NT-proBNP emerging as a particularly informative marker [18]. At the same time, proteomic patterns in exceptionally long-lived groups often reflect altered inflammatory tone and metabolic efficiency rather than uniform upregulation of classical “defense” proteins.
The Redox and Antioxidant “Paradox”
A recurring interpretive pitfall is to equate lower abundance of certain antioxidant-associated proteins with weaker defenses. In resilient aging phenotypes, lower expression of some stress-response proteins can reflect lower upstream oxidative burden, driven by more efficient mitochondria and reduced chronic inflammatory signaling. In other words, redox homeostasis may be achieved through reduced reactive species production rather than constant maximal antioxidant deployment. This framing aligns with mechanistic and epidemiologic literature linking oxidative stress pathways to cognitive outcomes and heterogeneity in brain aging trajectories [13].
Genetic Variants and Disease Delay
Genetic contributions to exceptional longevity appear to function largely through disease delay rather than prevention of aging itself. Among repeatedly implicated loci in long-lived populations are APOE (particularly APOE2 enrichment), FOXO3A variants associated with stress response and insulin signaling regulation, and lipid-related pathways including CETP-associated traits [22]. These variants plausibly support vascular and metabolic resilience, lowering the probability that age-related insults cross clinical thresholds.
Table 4. Key Molecular Mechanisms and Their Influence on Longevity
| Mechanism | Physiological impact | Role in longevity |
| Mitophagy | selective mitochondrial quality control | limits ROS from dysfunctional mitochondria; supports energetic resilience [23] |
| Proteostasis | balanced synthesis, folding, and degradation | reduces accumulation of aggregated proteins and organelle dysfunction [2,4] |
| Metabolic flexibility | switching between fuel sources | supports low baseline insulin and adaptive stress response [2,3] |
| Epigenetic stability | maintenance of gene regulation | may reduce maladaptive activation of growth/senescence programs [3] |
| Vascular compliance | preserved arterial elasticity | lowers wall stress and supports brain/heart microvascular function [15–17] |
Precision Geronutrition and Emerging Gerotherapeutics
As geroscience matures, the integration of nutritional strategy with pharmacologic interventions targeting hallmarks of aging has become a major frontier.
Targeted Pharmacologic Interventions (Evidence-Calibrated)
- Rapamycin/rapalogs: Among the most robust lifespan-extending interventions in animal models [1–3]. In humans, early clinical work suggests that mTOR inhibition can improve certain immune parameters in older adults, supporting geroscience relevance, though lifespan extension remains unproven [24].
- GLP-1 receptor agonists: Demonstrate strong cardiometabolic and cardiovascular outcome benefits in randomized trials in diabetes, with secondary relevance to aging biology via reductions in adiposity, inflammation, and cardiometabolic risk [25].
- SGLT2 inhibitors: Provide substantial reductions in heart failure hospitalization and cardiovascular mortality in randomized outcome trials, suggesting preservation of cardiovascular resilience; mechanistic links to mitochondrial energetics and inflammation are plausible, but claims of telomere lengthening or definitive geroprotection should be treated as investigational [26].
- Urolithin A: A gut microbiome–derived postbiotic that activates mitophagy pathways; randomized trial evidence indicates improvements in muscle performance and mitochondrial biomarker profiles in middle-aged adults, supporting a plausible role in mitochondrial quality control [27].
The Role of Caloric Restriction in Brain Health
The brain exhibits distinct vulnerabilities with age, including white matter and myelin integrity decline, microglial inflammatory shifts, and reduced metabolic resilience. High-resolution mapping studies in aging models indicate that caloric restriction can preserve region-specific gene programs linked to myelin maintenance, reduce expression of aging-associated inflammatory pathways, and support cellular states associated with neuroprotection [28]. While translating decades-long caloric restriction to humans is not straightforward, these findings support the concept that sustained metabolic moderation can preserve neurovascular and white matter biology relevant to cognitive resilience.
Regional Insights and Policy Impacts on Longevity
Longevity outcomes are not determined solely by biology; food environments and policy structures shape exposure to ultra-processed foods, dietary quality, and nutrition literacy. Public health tools that reduce decision friction may therefore have outsized effects on population-level cardiometabolic aging.
Front-of-Package Labeling and Nutrition Literacy
Randomized evidence indicates that front-of-package labeling systems—including label designs similar to the FDA’s proposed “Nutrition Info” concept—can improve consumer understanding and nudge healthier selection patterns in simulated purchasing tasks [29,30]. Effects can vary by nutrition literacy and sociodemographic factors; graded, spectrum-style labels may reduce disparities in comprehension by providing clearer comparative signals across products [29,30]. Such findings are relevant to longevity because small, sustained improvements in diet quality can compound over decades.
Oxidative Stress Vulnerability as a Precision Target
Individual vulnerability to oxidative stress and inflammatory signaling varies. Genetic and causal inference studies support a relationship between oxidative stress pathways and cognitive outcomes, suggesting that redox biology may contribute to heterogeneity in brain aging trajectories [13]. This reinforces a precision nutrition perspective: some individuals may benefit disproportionately from early optimization of cardiometabolic risk, dietary quality, and interventions that reduce chronic inflammation and oxidative burden.
Conclusion: A Lifetime Roadmap for Nutritional Longevity
Achieving exceptional longevity requires a strategic transition in nutritional priorities aligned to the biological realities of aging. Survival into the 11th decade appears supported by preserved mitochondrial quality control, maintained vascular compliance, and nutritional adequacy sufficient to overcome anabolic resistance despite declining appetite and physiologic reserve.
Recommended Nutritional Protocol for Longevity Transitions
- Early-to-mid life (20–65): Emphasize metabolic health and periodic downshifts in growth signaling (eg, caloric moderation or time-restricted eating). Maintain moderate protein intake and prioritize dietary patterns that support cardiometabolic health and lower chronic inflammation.
- Late-life transition (65–80): Increase protein toward ~1.0–1.2 g/kg/day as tolerated and clinically appropriate, with attention to per-meal distribution. Prioritize leucine-dense protein servings targeting ~2.5–3.0 g leucine per meal to support MPS signaling in anabolic resistance [6,7].
- Extreme old age (80–100+): Focus on nutrient density and feasible distribution across meals; appetite and chewing/swallowing constraints dominate. Monitor markers of cardiovascular stress (eg, NT-proBNP) and preserve vascular health through blood pressure control, physical activity where possible, and dietary patterns that support endothelial function.
Mitigating CVD and Dementia Risk
Maintaining vascular health through midlife remains essential to reduce late-life heart failure and cognitive vulnerability. CAC burden and arterial stiffness reflect cumulative exposure and risk, and their downstream consequences increasingly implicate brain aging as survival extends [12–17]. Preserving a physiologic mechanical environment—arterial elasticity and microvascular integrity—may reduce endothelial dysfunction and inflammatory amplification, supporting both cardiac and cognitive longevity.
In summary, the transition to longevity is not about doing “more” of the same interventions forever; it is a calculated pivot from growth suppression and maintenance prioritization in midlife to structural preservation and anabolic support in later life. By understanding nutrient sensing (notably leucine thresholds and mTOR dynamics) and the mechanobiology of vascular aging, individuals and clinicians can better navigate the biological hurdles separating standard geriatric aging from resilient centenarian trajectories.
References
- Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013;493(7432):338-345. doi:10.1038/nature11861
- Kennedy BK, Lamming DW. The Mechanistic Target of Rapamycin: The Grand ConducTOR of Metabolism and Aging. Cell Metab. 2016;23(6):990-1003. doi:10.1016/j.cmet.2016.05.009
- Papadopoli D, Boulay K, Kazak L, et al. mTOR as a central regulator of lifespan and aging. F1000Res. 2019;8:F1000 Faculty Rev-998. Published 2019 Jul 2. doi:10.12688/f1000research.17196.1
- Zarzycka W, Kobak KA, King CJ, Peelor FF 3rd, Miller BF, Chiao YA. Hyperactive mTORC1/4EBP1 signaling dysregulates proteostasis and accelerates cardiac aging. Geroscience. 2025;47(2):1823-1836. doi:10.1007/s11357-024-01368-w
- Breen L, Phillips SM. Skeletal muscle protein metabolism in the elderly: Interventions to counteract the ‘anabolic resistance’ of ageing. Nutr Metab (Lond). 2011;8:68. Published 2011 Oct 5. doi:10.1186/1743-7075-8-68
- Bauer J, Biolo G, Cederholm T, et al. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J Am Med Dir Assoc. 2013;14(8):542-559. doi:10.1016/j.jamda.2013.05.021
- Moore DR, Churchward-Venne TA, Witard O, et al. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J Gerontol A Biol Sci Med Sci. 2015;70(1):57-62. doi:10.1093/gerona/glu103
- Wolfson RL, Chantranupong L, Saxton RA, et al. Sestrin2 is a leucine sensor for the mTORC1 pathway. Science. 2016;351(6268):43-48. doi:10.1126/science.aab2674
- Saxton RA, Knockenhauer KE, Wolfson RL, et al. Structural basis for leucine sensing by the Sestrin2-mTORC1 pathway. Science. 2016;351(6268):53-58. doi:10.1126/science.aad2087
- Lixandrão ME, Longobardi I, Leitão AE, et al. Daily Leucine Intake Is Positively Associated with Lower Limb Skeletal Muscle Mass and Strength in the Elderly. Nutrients. 2021;13(10):3536. Published 2021 Oct 9. doi:10.3390/nu13103536
- Szwiega S, Pencharz PB, Rafii M, et al. Dietary leucine requirement of older men and women is higher than current recommendations. Am J Clin Nutr. 2021;113(2):410-419. doi:10.1093/ajcn/nqaa323
- Bos D, Vernooij MW, Elias-Smale SE, et al. Atherosclerotic calcification relates to cognitive function and to brain changes on magnetic resonance imaging. Alzheimers Dement. 2012;8(5 Suppl):S104-S111. doi:10.1016/j.jalz.2012.01.008
- Fan Z, Yang C, Qu X, et al. Association of Oxidative Stress on Cognitive Function: A Bidirectional Mendelian Randomisation Study. Mol Neurobiol. 2024;61(12):10551-10560. doi:10.1007/s12035-024-04231-3
- Huang GS, Hansen SL, McClelland RL, et al. Relation of Progression of Coronary Artery Calcium to Dementia (from the Multi-Ethnic Study of Atherosclerosis). Am J Cardiol. 2022;171:69-74. doi:10.1016/j.amjcard.2022.01.061
- Iadecola C. The pathobiology of vascular dementia. Neuron. 2013;80(4):844-866. doi:10.1016/j.neuron.2013.10.008
- Li Q, Qian Z, Huang Y, et al. Mechanisms of endothelial senescence and vascular aging. Biogerontology. 2025;26(4):128. Published 2025 Jun 25. doi:10.1007/s10522-025-10279-y
- Lai A, Zhou Y, Chheang C, et al. Decoding vascular aging: Substrate stiffness and shear stress orchestrate endothelial inflammation and remodelling via mechanosensitive pathways. Biomaterials. 2026;329:123932. doi:10.1016/j.biomaterials.2025.123932
- Hirata T, Arai Y, Yuasa S, et al. Associations of cardiovascular biomarkers and plasma albumin with exceptional survival to the highest ages. Nat Commun. 2020;11(1):3820. Published 2020 Jul 30. doi:10.1038/s41467-020-17636-0
- Levine ME, Suarez JA, Brandhorst S, et al. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014;19(3):407-417. doi:10.1016/j.cmet.2014.02.006
- Kurata H, Meguro S, Abe Y, et al. Dietary protein intake and all-cause mortality: results from The Kawasaki Aging and Wellbeing Project. BMC Geriatr. 2023;23(1):479. Published 2023 Aug 9. doi:10.1186/s12877-023-04173-w
- Budhathoki S, Sawada N, Iwasaki M, et al. Association of Animal and Plant Protein Intake With All-Cause and Cause-Specific Mortality in a Japanese Cohort. JAMA Intern Med. 2019;179(11):1509-1518. doi:10.1001/jamainternmed.2019.2806
- Brooks-Wilson AR. Genetics of healthy aging and longevity. Hum Genet. 2013;132(12):1323-1338. doi:10.1007/s00439-013-1342-z
- Shirakabe A, Ikeda Y, Sciarretta S, Zablocki DK, Sadoshima J. Aging and Autophagy in the Heart. Circ Res. 2016;118(10):1563-1576. doi:10.1161/CIRCRESAHA.116.307474
- Mannick JB, Del Giudice G, Lattanzi M, et al. mTOR inhibition improves immune function in the elderly. Sci Transl Med. 2014;6(268):268ra179. doi:10.1126/scitranslmed.3009892
- Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2016;375(4):311-322. doi:10.1056/NEJMoa1603827
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373(22):2117-2128. doi:10.1056/NEJMoa1504720
- Singh A, D’Amico D, Andreux PA, et al. Urolithin A improves muscle strength, exercise performance, and biomarkers of mitochondrial health in a randomized trial in middle-aged adults. Cell Rep Med. 2022;3(5):100633. doi:10.1016/j.xcrm.2022.100633
- Zhang Z, Epstein A, Schaefer C, et al. Spatiotemporal profiling reveals the impact of caloric restriction in the aging mammalian brain. Cell Rep. 2025;44(9):116165. doi:10.1016/j.celrep.2025.116165
- Grummon AH, O’Sullivan K, Petimar J, et al. Nutrition Info and Other Front-of-Package Labels and Simulated Food and Beverage Purchases: A Randomized Clinical Trial. JAMA Netw Open. 2025;8(10):e2537389. Published 2025 Oct 1. doi:10.1001/jamanetworkopen.2025.37389
- Huang Y, O’Sullivan K, Block JP, Petimar J, Lee CJY, Grummon AH. Impact of the Food and Drug Administration’s Proposed Front-of-Package Label and Alternative Designs on Consumer Understanding: A Randomized Experiment. Am J Prev Med. Published online December 13, 2025. doi:10.1016/j.amepre.2025.108222


