The Surprising Truth About Exercise and Clogged Arteries
Introduction: The Heart of the Matter
Many people feel a deep fear when they hear the words “clogged arteries.” We often think of our heart like the plumbing in an old house. We imagine that over time, the pipes get filled with thick gunk. In this simple view, if the pipes are clear, you are healthy. If the pipes are full of gunk, you are in big trouble. We assume that people who exercise a lot will have the cleanest pipes of all. But science is showing us a very strange and surprising truth. It is called the “Athlete’s Paradox.” Researchers have found that people who exercise the most—like lifelong runners or cyclists—sometimes have much more plaque in their heart than people who sit on the couch.
If you just look at a heart scan, it might look like the athlete is in danger. However, I want to show you why this is not as scary as it sounds. Your heart is much smarter than a metal pipe. It can change its shape and its chemistry to stay safe. This news is actually very encouraging for anyone who loves to move. Let’s look at why your heart scan might be lying to you.
Takeaway 1: The “Expanding Pipe” Trick (Vascular Remodeling)
In the past, doctors looked at the heart using a “lumenogram.” This is like taking a picture of the water flowing through a pipe. If the water path looks wide, doctors thought the pipe was clean. However, a famous scientist named Dr. Glagov proved that this doesn’t tell the whole story. He discovered something called the “Glagov phenomenon.”
Imagine a garden hose. If dirt starts building up on the inside walls, you would expect the water path to get smaller right away. But our arteries are alive and very clever. They perform a trick called “positive remodeling.” When gunk builds up, the artery actually stretches and grows larger on the outside. This keeps the water path on the inside wide and clear. Dr. Glagov found that if the buildup is between 0 and 40 percent, the inside path doesn’t shrink at all! A scan might show a “normal” pipe, even if there is a lot of plaque hidden inside the walls.
Exercise is the “master regulator” of this stretching process. Constant movement and fast-flowing blood tell your arteries to expand. This means that even if an athlete has some plaque, their “pipes” have grown so large that the blood can still zoom through without any problems. The vessel adapts to the gunk by making more room.
Takeaway 2: HIIT Can Be as Strong as Medicine
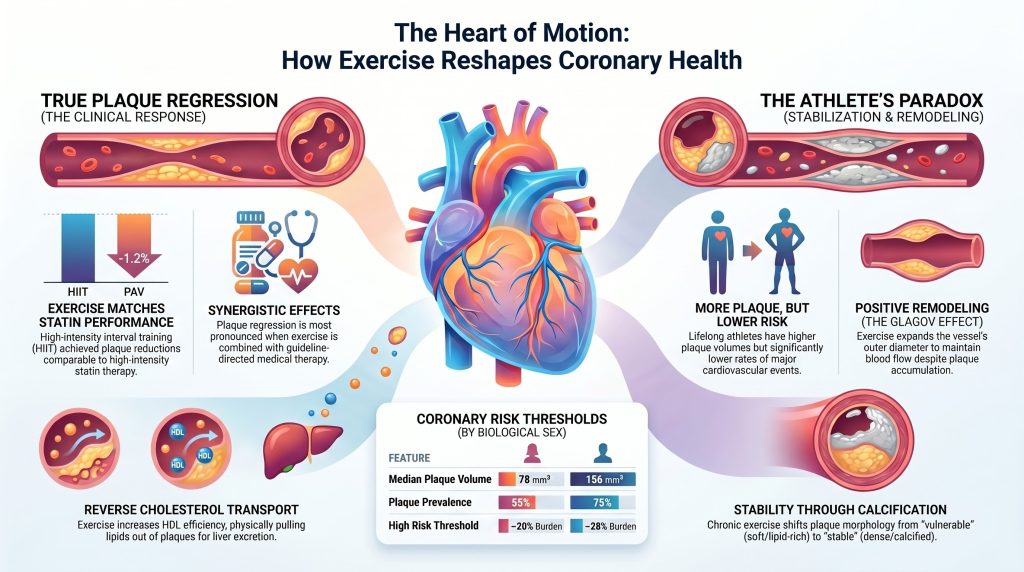
We usually think of statins, which are common cholesterol pills, as the only way to fix our arteries. Medicine is very good at its job. On average, these pills can reduce the amount of plaque by about 1.1 percent. But new research called the CENIT trial shows that high-intensity exercise can be just as strong as medicine.
The CENIT trial looked at people who already had heart disease. They put one group through High-Intensity Interval Training (HIIT). This means doing short bursts of very hard work, like running fast for a minute and then walking for a minute. They found that HIIT reduced plaque volume by 1.2 percent. It also reduced the total amount of “gunk” (called TAVnorm) by a huge 12.0 cubic millimeters. This is amazing because it shows exercise can work just as well as—or even better than—a pill.
The Comparison: Standard Care vs. Adding HIIT
- Before (Standard Medicine Only): Plaque is reduced by about 1.1 percent. This is good, but the “pipes” stay mostly the same size.
- After (Medicine PLUS HIIT Exercise): Plaque is reduced by 1.2 percent, and the total volume of gunk drops significantly. The heart becomes much cleaner.
The most important part is that these people were already taking their heart medicine. The HIIT was a “bonus” that made their hearts even healthier. As the researchers noted:
“The CENIT results are notable because they demonstrate that exercise can achieve plaque reductions of a magnitude comparable to medicine, even in people already receiving top-tier medical care.”
Takeaway 3: The Athlete’s Paradox (More Plaque, Less Risk)
A study called Master@Heart looked at lifelong athletes and compared them to healthy people who did not exercise much. You would think the athletes would have the cleanest hearts. But the data showed something very weird. Lifelong athletes actually had more plaque and higher “calcium scores” than the people who sat still.
Scientists use “Odds Ratios” (OR) to explain risk. If a score is 1.0, the risk is normal. If it is higher than 1.0, the risk is higher. Lifelong athletes had an OR of 1.86 for having any plaque. They had an OR of 1.96 for having plaque in the most important “proximal” parts of the heart. In simple terms, these athletes were nearly twice as likely to have plaque as the non-athletes.
This sounds like bad news, but it isn’t! Even though these athletes have more plaque on their scans, they have fewer heart attacks and live much longer. This is the “Athlete’s Paradox.” The scan shows more “gunk,” but the person is actually much safer. The amount of plaque on a screen doesn’t tell you how strong the heart really is.
Takeaway 4: Soft Plaque vs. Hard Shells
Why are athletes safer if they have more plaque? It is because of the type of plaque they have. There are two main kinds: “vulnerable” and “stable.” Vulnerable plaque is soft and filled with fat. This is the dangerous kind that can pop like a pimple and cause a heart attack. Stable plaque is hard and “calcified.”
Think of calcification like a “scab” inside your artery. When you scrape your knee, your body makes a hard scab to protect the skin while it heals. Your heart does the same thing. It takes soft, dangerous plaque and covers it in a hard shell. This makes the plaque “stable” so it won’t pop.
We must be honest about the data: the Master@Heart study showed athletes actually have higher odds of having soft plaque too (an OR of 2.80). However, the total effect of exercise is to turn the heart into a fortress. Even if they have more soft plaque, they have even more hard plaque to balance it out. The study explains:
“The net effect in chronically active individuals is a transition from compositionally vulnerable to compositionally stable plaque morphology.”
Exercise helps the body “heal” the arteries by turning the gunk into hard, safe shells.
Takeaway 5: Why Speed Matters More than Distance (The 9 MET Threshold)
How hard you push yourself changes how your heart looks. A study called MARC-2 looked at “exercise volume” (how long you work out) versus “exercise intensity” (how hard you work).
Scientists measure intensity using “METs.”
- Vigorous Exercise (6 to 9 METs): This is like a fast walk or a light jog. This level was actually linked to less plaque growth. It slowed down the “rusting” of the pipes.
- Very Vigorous Exercise (Over 9 METs): This is “redlining” the engine. It is pushing your heart to the absolute limit. This was linked to more plaque and more calcification.
Think of it like a car. Driving at a steady, fast speed is great for the engine. It keeps everything running smooth. But if you push the car as fast as it can possibly go every single day, the engine will start to show some wear and tear. For athletes, pushing very hard makes the plaque build up and harden faster. This isn’t necessarily bad, but it explains why the scans of very fast runners look different than those of casual walkers.
Takeaway 6: The “Small Pipe” Problem for Women
Heart health is not the same for everyone. A study called the PROMISE trial found that women’s hearts face a unique challenge. The reason is simple: women usually have smaller coronary arteries than men.
Because the “pipes” are smaller, even a little bit of gunk takes up more space. Imagine a small pebble. If you drop that pebble into a wide drainpipe, the water still flows fine. But if you drop that same pebble into a thin drinking straw, the straw gets blocked.
- For Men: Their risk starts to go up when plaque fills about 42% of the vessel.
- For Women: Their risk starts to go up much earlier, at only 32%.
This means the “safe ceiling” for plaque is lower for female athletes. A woman might have less total plaque than a man, but it might be more dangerous for her because her vessels are smaller. Female athletes need to be extra careful and keep an eye on their heart health sooner than men do.
Takeaway 7: It’s Not Just Your Zip Code or Your Partner
We often think our health is decided by where we live or who we marry. It is true that couples share a lot. In fact, 95% of partners share the same eating habits! If your partner eats junk food, you probably do too. Also, people with more money or better jobs often have better heart health.
But your own choice to move is more powerful than your environment. We know this because of a famous study of 31,000 London transport workers. Researchers compared bus drivers to bus conductors. These men were the same age, had the same pay, and lived in the same neighborhoods. But the drivers sat in a seat all day. The conductors walked up and down the stairs of the double-decker buses hundreds of times every shift.
The results were shocking. The conductors—the ones who moved—had half the number of heart attacks as the drivers. This shows that movement itself is the magic ingredient. Even if you live in a certain “zip code” or share a kitchen with someone who doesn’t eat well, your choice to be active can rewrite the future of your heart.
Takeaway 8: The Body’s Internal “Garbage Trucks”
Exercise doesn’t just change the shape of your heart; it changes the chemistry. Your body has a special cleanup crew called “Reverse Cholesterol Transport.”
Think of HDL particles as “garbage trucks.” Their job is to drive through your arteries and pick up the “trash” (fat and cholesterol). Then they take that trash to the “dump” (your liver) to be thrown away. Exercise makes these garbage trucks much better at their job. It gives you more trucks and makes them faster at picking up the trash.
Movement also creates “Wall Shear Stress.” This is the friction of blood rubbing against the walls of your arteries. While friction sounds like it might be bad, this specific kind is very good. It triggers a chemical called “nitric oxide.” I like to think of nitric oxide as a magic spray that keeps the pipe walls slippery and relaxed. It stops inflammation and makes it very hard for “gunk” to stick to the walls. It’s like having a self-cleaning oven inside your chest!
Conclusion: A New Way to Look at Your Heart
For a long time, people thought that any plaque on a heart scan was a sign of failure. We believed that the goal of exercise was to keep our arteries as clean and shiny as a brand-new metal pipe. But modern science has shown us a much more hopeful and interesting story. Your heart is a living, breathing thing that knows how to protect itself.
The “Athlete’s Paradox” teaches us that the amount of plaque on a scan is not the most important number. What matters more is the type of plaque and the strength of the vessel. If you exercise, your heart learns to grow wider. It learns to turn soft, dangerous gunk into hard, stable “scabs” that won’t cause trouble. It builds a better cleanup crew of garbage trucks and keeps its walls slippery and healthy.
Instead of worrying about a single number on a scan, we should focus on “functional reserve.” This is a fancy way of asking: “What can your heart actually do?” Is it strong? Can it handle a tough workout? Does it have the wide, flexible pipes that only movement can provide? The real question isn’t “How much plaque do I have?” but rather: “How well-built is my heart for the long journey ahead?”
DEEP DIVE
Chronic Exercise and the Structural Evolution of Coronary Atheroma: A Critical Appraisal of Regression, Remodeling, and the Athlete’s Paradox
The quest to determine whether physical exertion can fundamentally reverse the progression of coronary artery disease has transitioned from early epidemiological observations to a highly technical discourse centered on the micro-architecture of the vessel wall. In the modern era of cardiology, characterized by rapid advancements in artificial intelligence–enabled quantitative coronary computed tomography angiography (AI-QCTA) and high-resolution intravascular imaging, the traditional metric of luminal stenosis is increasingly viewed as an insufficient silhouette of a far more complex biological process [1]. The fundamental tension in preventive cardiology today lies in whether the undisputed cardiovascular benefits of exercise are realized through the actual reduction of plaque mass — true regression — or whether exercise serves as a master regulator of vascular remodeling and plaque stabilization, effectively mitigating the risk of clinical events without necessarily eliminating the underlying disease burden [2].
This report critically evaluates the evidence surrounding this tension, prioritizing high-fidelity imaging data from intravascular ultrasound (IVUS), optical coherence tomography (OCT), and coronary computed tomography angiography (CCTA). By synthesizing data from matched cohorts, longitudinal athlete registries, and mechanistic models of shear stress, this analysis tests the hypothesis that exercise primarily functions as a stabilizer and remodeler of the coronary tree rather than a primary agent of plaque regression.
Plaque Volume versus Lumen Size: The Glagovian Context
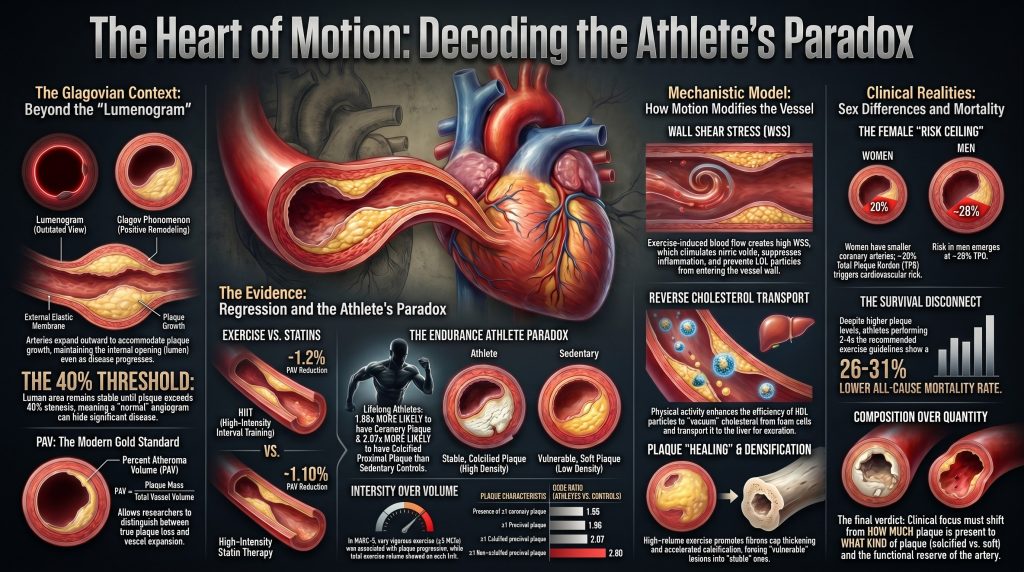
The accurate assessment of exercise-induced changes in the coronary arteries requires a nuanced understanding of the relationship between the vessel’s external boundaries and its internal lumen. For decades, the gold standard for diagnosing coronary artery disease was invasive coronary angiography, which provides a two-dimensional map of the lumen — a “lumenogram.” However, this approach is fundamentally limited by the Glagov phenomenon, or positive remodeling, wherein the coronary artery undergoes a progressive enlargement of its external elastic membrane (EEM) in response to the accumulation of plaque [3].
This compensatory expansion allows the vessel to maintain its luminal cross-sectional area even as a significant atheroma develops. In Glagov’s seminal histologic study of 136 autopsied left main coronary arteries, lumen area did not decrease as a function of percent stenosis between 0 and 40 percent, but diminished markedly above that threshold (r = −0.73, P < 0.001) [3]. This implies that an angiographically normal vessel may harbor a substantial, and potentially vulnerable, plaque burden. The PROSPECT IVUS sub-analysis confirmed in vivo that EEM cross-sectional areas increase in proportion to plaque-and-media area in 552 left main coronary arteries (r = 0.61, P < 0.0001 overall, rising to r = 0.88 when plaque burden was at or below 20 percent) [4].
Exercise is a potent stimulus for vascular remodeling. Chronic aerobic training induces sustained increases in blood flow and shear stress, which activate endothelial pathways that increase nitric-oxide bioavailability and the structural caliber of conduit arteries [5]. Consequently, any study claiming that exercise reduces atherosclerosis based solely on luminal diameter is likely measuring a combination of actual plaque loss and compensatory vessel expansion. To distinguish between these effects, researchers rely on percent atheroma volume (PAV), which normalizes plaque mass to the total volume of the vessel. PAV is calculated as the sum of (EEM area − lumen area) divided by the sum of EEM area, expressed as a percentage. This metric ensures that if a vessel grows larger (increased EEM) but the plaque area remains constant, PAV decreases, reflecting a relative reduction in the occluded proportion of the artery, even when absolute total atheroma volume (TAV) remains stable [6].
Evidence for True Plaque Regression: Benchmarking against Pharmacotherapy
When evaluating the capacity of exercise to induce regression, it is necessary to establish a benchmark using lipid-lowering therapies (LLTs), which have the most robust data for reducing PAV and TAV. A comprehensive meta-analysis of 51 studies involving 9,113 adults demonstrated that LLTs as a class reduce PAV by a mean of −1.10 percent (95% CI −1.63 to −0.56, P < 0.01) and reduce TAV by −5.84 mm³ (95% CI −8.64 to −3.04, P < 0.01); high-intensity statins drove the largest TAV effect (−7.60 mm³; 95% CI −11.89 to −3.31, P < 0.01) [6]. Direct head-to-head plaque-imaging evidence from the GLAGOV randomized trial further demonstrates that PCSK9 inhibition added to statin therapy produces incremental PAV regression in patients with established coronary artery disease [2].
In comparison, the direct evidence for exercise-induced regression is emerging but remains modest. The CENIT trial (Coronary Disease and the Effect of High-Intensity Interval Training) provided critical longitudinal data by randomizing 60 patients with stable coronary artery disease, all of whom underwent percutaneous coronary intervention, to either six months of supervised high-intensity interval training (HIIT) at 85–95 percent of peak heart rate or to contemporary preventive guidelines [7]. The study used IVUS to measure changes in PAV and TAV in residual coronary segments.
| Parameter | HIIT Group (n=30) | Control Group (n=30) | Between-Group Difference |
| Change in PAV (%) | −1.2 (95% CI: −2.1 to −0.2; P=0.017) | +0.2 (95% CI: −0.7 to 1.1; P=0.616) | −1.4 (95% CI: −2.7 to −0.1; P=0.036) [7] |
| Change in TAVnorm (mm³) | −9.0 (95% CI: −14.7 to −3.4; P=0.002) | — | −12.0 (95% CI: −19.9 to −4.2; P=0.003) [7] |
The CENIT results are notable because they demonstrate that exercise can achieve PAV reductions of a magnitude comparable to lipid-lowering therapy (−1.2% vs. −1.10%) [6], [7]. Importantly, this regression was observed in a population already receiving guideline-directed medical therapy, making it difficult to isolate the independent contribution of exercise from a synergistic interaction with statins.
The Endurance Athlete Paradox
While interventional trials in patients with established coronary artery disease suggest the potential for regression, observational data from healthy veteran athletes present a challenging paradox. Large registries, including MARC (Measuring Athlete’s Risk of Cardiovascular Events) and Master@Heart, consistently report that lifelong endurance athletes have a higher prevalence of coronary atherosclerotic plaque and higher Coronary Artery Calcium (CAC) scores than less active individuals with comparable cardiovascular risk profiles [8], [9].
The Master@Heart study, a prospective observational cohort analysis of 191 lifelong master endurance athletes, 191 late-onset athletes (initiating endurance sports after age 30), and 176 healthy non-athletic controls (all male, low cardiovascular risk profile, median age 55 years), reported the following adjusted associations for lifelong endurance sports relative to a healthy non-athletic lifestyle [9]:
- ≥1 coronary plaque: OR 1.86 (95% CI 1.17–2.94)
- ≥1 proximal plaque: OR 1.96 (95% CI 1.24–3.11)
- ≥1 calcified plaque: OR 1.58 (95% CI 1.01–2.49)
- ≥1 calcified proximal plaque: OR 2.07 (95% CI 1.28–3.35)
- ≥1 non-calcified plaque: OR 1.95 (95% CI 1.12–3.40)
- ≥1 non-calcified proximal plaque: OR 2.80 (95% CI 1.39–5.65)
- ≥1 mixed plaque: OR 1.78 (95% CI 1.06–2.99)
The finding of higher odds of non-calcified (lipid-rich) plaques in proximal segments is particularly notable, as such plaques are typically associated with higher near-term risk than purely calcified lesions [9].
The MARC-2 study added longitudinal granularity by distinguishing exercise volume from intensity in 289 middle-aged and older male athletes (median age 54 years) followed for a mean of 6.3 years [8]. Total exercise volume (MET-hours/week) showed no association with progression of CAC or plaque. By contrast, exercise intensity stratified by metabolic equivalents — vigorous (6–9 METs) versus very vigorous (≥9 METs) — was significantly associated with imaging outcomes:
| Exercise Intensity | Effect on CAC Score (β per 10% increase) | Plaque Progression |
| Vigorous (6–9 METs) | −0.05 (95% CI −0.09 to −0.01; P=0.02) [8] | Less progression |
| Very Vigorous (≥9 METs) | +0.05 (95% CI 0.01 to 0.09; P=0.01) [8] | aOR 1.09 (95% CI 1.01–1.18) per 10% increase [8] |
| Total Volume | No significant association [8] | No significant association [8] |
That intensity rather than volume drives plaque and CAC progression in athletes suggests a physiological threshold beyond which exercise may exert pro-atherogenic effects. Proposed mechanisms include exercise systolic blood pressures that can exceed 200 mmHg in well-trained athletes during near-maximal effort [25], repetitive mechanical stress on the coronary wall, and inflammatory signaling pathways implicated in athlete-related vascular calcification [10].
The defining feature of the athlete’s paradox, however, is that despite higher plaque burden, endurance athletes have lower rates of major adverse cardiovascular events (MACE) than the general population. Although the rank order of plaque morphology (calcified > mixed > non-calcified) is broadly similar across active and sedentary populations, lifelong endurance athletes who harbor plaque are more likely than sedentary controls to harbor exclusively calcified lesions, which are considered more stable [12]. This phenotypic enrichment for dense, calcified (stable) lesions — rather than for mixed or non-calcified vulnerable lesions — suggests that high-volume exercise may accelerate the natural healing or densification process of atherosclerosis [10], [11].
Matched Cohort Evidence and Socioeconomic Confounders
A critical requirement for validating the impact of exercise is the control of social and economic confounders. Higher socioeconomic status (SES) is independently associated with both greater exercise capacity and improved cardiovascular outcomes [13]. In the Heart and Soul Study, an analysis of 943 men and women with stable coronary heart disease, exercise capacity decreased in a graded fashion across categories of income, education, housing, and occupation. After multivariable adjustment, the difference in exercise capacity between the highest and lowest categories was 2.4 METs for household income, 1.8 METs for education, 2.3 METs for housing, and 1.3 METs for occupation (P < 0.001 for all trends) [13].
Spousal Concordance and Partner Studies
Cohabiting partners share a micro-environment encompassing diet, socioeconomic stress, and access to healthcare. In a US nationwide cohort of 5,364 married or domestic-partner couples, within-couple concordance exceeded 50 percent for every component of the American Heart Association’s Life’s Simple 7 framework, ranging from 53 percent for total cholesterol to 95 percent for healthy diet score; concordance for metabolic syndrome reached 73 percent [14]. Despite this strong concordance for behaviors and risk factors, marital status itself, when adjusted for cardiovascular risk factors, was not independently associated with subclinical coronary atherosclerosis on CCTA in a study of 9,288 asymptomatic individuals [15]. Together, these findings suggest that the shared environment of a partnership produces a background risk level, but the trajectory of plaque burden is more heavily influenced by individual health behaviors — including the choice to exercise regularly — than by partnership status itself.
Occupational Activity as a Controlled Environment
The London Transport Workers Study remains the canonical example of an occupational analog [16]. By comparing conductors and drivers within the same transport system — men of similar age and social class — Jeremy Morris was able to isolate occupational physical activity from most confounders. Morris and colleagues studied tens of thousands of London bus and postal workers and reported that physically active conductors, who climbed and descended hundreds of stairs per shift, experienced approximately half the rate of acute and sudden coronary deaths observed among the sedentary drivers [16], [17]. This natural experiment provides some of the earliest robust evidence that occupational physical activity independently modifies the clinical expression of coronary disease, even when the macro-environment is held constant.
Dose-Response Relationships and the Mortality Curve
The relationship between exercise dose and mortality is widely regarded as curvilinear, with the most substantial risk reductions occurring as individuals move from sedentary to moderately active. The systematic review and meta-analysis by Blond and colleagues, encompassing 48 prospective cohort studies, used random-effects restricted cubic spline dose-response modeling to estimate hazard ratios across the full exposure range. Compared with the recommended physical activity dose (750 MET-min/week), risk continued to decrease beyond recommendations: at 5,000 MET-min/week, hazard ratios were approximately 0.86 for all-cause mortality and 0.73 for cardiovascular mortality. No excess risk was observed at the highest exposure levels examined [18].
The Lee et al. Circulation analysis of 116,221 adults pooled from the Nurses’ Health Study and the Health Professionals Follow-up Study, with up to 30 years of follow-up and approximately 47,000 deaths, examined long-term physical activity intensity in detail [19]. Compared with no leisure-time vigorous physical activity (VPA), participants meeting the guideline (75–149 min/week of VPA) had hazard ratios of 0.81 (95% CI 0.76–0.87) for all-cause mortality, 0.69 (95% CI 0.60–0.78) for CVD mortality, and 0.85 (95% CI 0.79–0.92) for non-CVD/non-cancer mortality. Participants performing two to four times the recommended minimum had a 21–23 percent lower all-cause mortality for VPA (150–299 min/week) and a 26–31 percent lower all-cause mortality for moderate physical activity (300–599 min/week) [19]. Higher long-term doses (≥300 min/week VPA or ≥600 min/week moderate activity) did not provide further mortality reduction, but no excess risk was observed [19].
This mortality benefit persists despite the athlete’s paradox of increased CAC, supporting the conclusion that the plaques found in highly active individuals are compositionally more benign in their clinical manifestation [10], [11].
Mechanistic Model: How Exercise Modifies Plaque
The biological mechanisms by which exercise potentially reduces plaque burden or facilitates stabilization act both systemically and locally.
Fluid Mechanics and Wall Shear Stress
Plaque development is governed by patterns of blood flow. Laminar flow produces high wall shear stress (WSS), which maintains endothelial homeostasis through the mechanosensitive transcription-factor axis (KLF2 and KLF4) and downstream activation of endothelial nitric oxide synthase. Disturbed or oscillatory flow, common at coronary bifurcations, produces low WSS that promotes an atherogenic endothelial phenotype, including upregulation of NF-κB-driven adhesion molecules, monocyte recruitment, and increased endothelial transcytosis of LDL [20]. Exercise-induced increases in flow rate directly enhance the protective frictional force at the vessel wall. Direct molecular evidence in human coronary arteries has shown that regular physical activity in patients with coronary artery disease increases phosphorylation of endothelial nitric oxide synthase at Ser1177, the key regulatory site governing nitric oxide production [5].
Reverse Cholesterol Transport and HDL Functionality
Exercise facilitates the removal of lipids from plaque via the reverse cholesterol transport pathway, beginning with cholesterol efflux from foam cells to HDL particles, which transport lipids to the liver for hepatobiliary excretion. A randomized exercise-dosing trial measuring multiple HDL functional metrics has shown that increasing both exercise intensity and dose produces dose-responsive improvements in HDL cholesterol efflux capacity, anti-oxidant capacity, and anti-inflammatory function — effects that are largely independent of changes in HDL cholesterol concentration alone [22].
Plaque Stabilization, Matrix Metalloproteinases, and the Fibrous Cap
Beyond regression, exercise promotes structural stabilization through fibrous-cap thickening, mediated in part by reduced intra-plaque matrix metalloproteinase activity. Animal studies in apolipoprotein E knockout mice show that aerobic exercise training reduces intra-plaque MMP-8 and MMP-9 activity, increases tissue inhibitor of metalloproteinases (TIMP-1, TIMP-2), increases fibrous-cap thickness, and reduces the relative size of the necrotic lipid core [23]. Human systematic-review evidence links chronic physical activity to favorable changes in circulating MMP-9, oxidized LDL, and inflammatory mediators relevant to plaque vulnerability [21]. Concurrent reductions in systemic inflammation, including C-reactive protein, may further contribute to plaque stabilization [11], [21]. The net effect in chronically active individuals is a transition from compositionally vulnerable to compositionally stable plaque morphology, even when total plaque volume increases [10], [12].
Sex-Specific Differences in Plaque Risk
A crucial refinement in modern preventive cardiology is the recognition that plaque burden carries different prognostic implications in men and women. In a recent analysis of the PROMISE trial CCTA arm (4,267 patients, approximately 51 percent women, median follow-up 26 months), women had lower coronary plaque prevalence than men (55 percent versus 75 percent; P < 0.001) and lower median total plaque volume, yet a similar incidence of major adverse cardiovascular events (2.3 percent versus 3.4 percent) [24]. Critically, when MACE risk was modeled against total plaque burden using sex-stratified spline Cox regression, the hazard ratio crossed unity at a substantially lower plaque-burden threshold in women than in men.
| Feature | Women | Men | Implication |
| Plaque prevalence | 55% | 75% | Lower prevalence in women [24] |
| Plaque volume | Lower (median) | Higher (median) | Smaller absolute burden in women [24] |
| Total plaque burden at HR=1.0 | ≈20% | ≈28% | Risk emerges at a lower burden in women [24] |
| Total plaque burden at HR=1.5 | ≈32% | ≈42% | Steeper risk trajectory in women [24] |
These data indicate that uniform thresholds for high-risk plaque underestimate risk in women: because women have smaller coronary arteries, a smaller absolute plaque volume produces a larger relative plaque burden and a more rapid acceleration of MACE risk [24]. This implies that for female athletes, the ceiling for compositionally benign subclinical plaque may be considerably lower than for their male counterparts.
Testing the Hypothesis: Remodeling or Regression?
The hypothesis under examination is that exercise does not meaningfully reduce total coronary plaque burden but rather improves vessel caliber and plaque stability. The evidence supports a population-dependent answer:
In patients with established CAD on guideline-directed therapy: Exercise can meaningfully reduce total plaque burden. The CENIT trial demonstrated a between-group PAV difference of −1.4 percent and a TAVnorm reduction of −12 mm³ after six months of HIIT [7].
In healthy lifelong endurance athletes: Exercise does not reduce total plaque burden but is associated with higher prevalence and burden across all plaque types [9]. In this population, the hypothesis is largely supported: the cardiovascular benefit of exercise is realized through vascular remodeling and stabilization (calcification) rather than removal.
Apparent versus true benefits: Increased lumen size in trained individuals reflects a real physiological adaptation (positive remodeling) that confers significant clinical protection through enhanced coronary flow reserve [3], [4]. This remodeling benefit is distinct from the discrete benefit of true plaque regression, and the two effects operate on different timescales.
Final Verdict: Regression, Progression, or Stabilization?
The totality of the evidence indicates that chronic exercise is a multi-modal modifier of coronary atherosclerosis. True regression — measurable reductions in PAV and TAV — is achievable through high-intensity exercise interventions, particularly in populations with existing coronary artery disease, where the magnitude approaches that of intensive lipid-lowering therapy [2], [6], [7]. Among individuals without known disease, moderate exercise appears to slow the progression of soft, vulnerable, lipid-rich plaques through favorable lipid metabolism, enhanced HDL cholesterol efflux capacity, and reduced systemic inflammation [21], [22]. In high-volume endurance athletes, exercise functions primarily as a stabilizing agent, paradoxically accelerating calcification while expanding vessel caliber through positive remodeling — yielding a compositionally benign atherosclerotic burden that does not translate into elevated clinical risk [9], [10], [12].
Critically, the survival benefit of exercise is decoupled from absolute plaque volume: extreme athletes have more plaque yet exhibit substantially lower all-cause and CVD mortality than the general population [18], [19]. The clinical emphasis should therefore shift from quantifying how much plaque is present toward characterizing what kind of plaque is present and what functional reserve the individual’s coronary tree retains.
References
- Narula J, Stuckey TD, Nakazawa G, et al. Prospective deep learning-based quantitative assessment of coronary plaque by computed tomography angiography compared with intravascular ultrasound: the REVEALPLAQUE study. Eur Heart J Cardiovasc Imaging. 2024;25(9):1287-1295. doi:10.1093/ehjci/jeae115
- Nicholls SJ, Puri R, Anderson T, et al. Effect of Evolocumab on Progression of Coronary Disease in Statin-Treated Patients: The GLAGOV Randomized Clinical Trial. JAMA. 2016;316(22):2373-2384. doi:10.1001/jama.2016.16951
- Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ. Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med. 1987;316(22):1371-1375. doi:10.1056/NEJM198705283162204
- Inaba S, Mintz GS, Shimizu T, et al. Compensatory enlargement of the left main coronary artery: insights from the PROSPECT study. Coron Artery Dis. 2014;25(2):98-103. doi:10.1097/MCA.0000000000000074
- Hambrecht R, Adams V, Erbs S, et al. Regular physical activity improves endothelial function in patients with coronary artery disease by increasing phosphorylation of endothelial nitric oxide synthase. Circulation. 2003;107(25):3152-3158. doi:10.1161/01.CIR.0000074229.93804.5C
- Rivera FB, Cha SW, Varona MC, et al. Atherosclerotic coronary plaque regression from lipid-lowering therapies: A meta-analysis and meta-regression. Am J Prev Cardiol. 2024;18:100645. Published 2024 Mar 11. doi:10.1016/j.ajpc.2024.100645
- Vesterbekkmo EK, Aksetøy IA, Follestad T, et al. High-intensity interval training induces beneficial effects on coronary atheromatous plaques: a randomized trial. Eur J Prev Cardiol. 2023;30(5):384-392. doi:10.1093/eurjpc/zwac309
- Aengevaeren VL, Mosterd A, Bakker EA, et al. Exercise Volume Versus Intensity and the Progression of Coronary Atherosclerosis in Middle-Aged and Older Athletes: Findings From the MARC-2 Study. Circulation. 2023;147(13):993-1003. doi:10.1161/CIRCULATIONAHA.122.061173
- De Bosscher R, Dausin C, Claus P, et al. Lifelong endurance exercise and its relation with coronary atherosclerosis. Eur Heart J. 2023;44(26):2388-2399. doi:10.1093/eurheartj/ehad152
- Hsu JJ, Tintut Y, Demer LL. Paradox of Exercise and Coronary Artery Calcification: Potential Underlying Mechanisms. Circ Res. 2025;137(2):335-349. doi:10.1161/CIRCRESAHA.125.326011
- Aengevaeren VL, Mosterd A, Sharma S, et al. Exercise and Coronary Atherosclerosis: Observations, Explanations, Relevance, and Clinical Management. Circulation. 2020;141(16):1338-1350. doi:10.1161/CIRCULATIONAHA.119.044467
- Aengevaeren VL, Mosterd A, Braber TL, et al. Relationship Between Lifelong Exercise Volume and Coronary Atherosclerosis in Athletes. Circulation. 2017;136(2):138-148. doi:10.1161/CIRCULATIONAHA.117.027834
- Cohen B, Vittinghoff E, Whooley M. Association of socioeconomic status and exercise capacity in adults with coronary heart disease (from the Heart and Soul Study). Am J Cardiol. 2008;101(4):462-466. doi:10.1016/j.amjcard.2007.09.093
- Shiffman D, Louie JZ, Devlin JJ, Rowland CM, Mora S. Concordance of Cardiovascular Risk Factors and Behaviors in a Multiethnic US Nationwide Cohort of Married Couples and Domestic Partners. JAMA Netw Open. 2020;3(10):e2022119. Published 2020 Oct 1. doi:10.1001/jamanetworkopen.2020.22119
- Ann SH, Lee H, Park KS, et al. Marital Status and Subclinical Coronary Atherosclerosis in Asymptomatic Individuals. J Am Heart Assoc. 2022;11(11):e024942. doi:10.1161/JAHA.121.024942
- Morris JN, Heady JA, Raffle PA, Roberts CG, Parks JW. Coronary heart-disease and physical activity of work. Lancet. 1953;262(6795):1053-1057. doi:10.1016/s0140-6736(53)90665-5
- Morris JN, Kagan A, Pattison DC, Gardner MJ. Incidence and prediction of ischaemic heart-disease in London busmen. Lancet. 1966;2(7463):553-559. doi:10.1016/s0140-6736(66)93034-0
- Blond K, Brinkløv CF, Ried-Larsen M, Crippa A, Grøntved A. Association of high amounts of physical activity with mortality risk: a systematic review and meta-analysis. Br J Sports Med. 2020;54(20):1195-1201. doi:10.1136/bjsports-2018-100393
- Lee DH, Rezende LFM, Joh HK, et al. Long-Term Leisure-Time Physical Activity Intensity and All-Cause and Cause-Specific Mortality: A Prospective Cohort of US Adults. Circulation. 2022;146(7):523-534. doi:10.1161/CIRCULATIONAHA.121.058162
- Niu N, Xu S, Xu Y, Little PJ, Jin ZG. Targeting Mechanosensitive Transcription Factors in Atherosclerosis. Trends Pharmacol Sci. 2019;40(4):253-266. doi:10.1016/j.tips.2019.02.004
- Palmefors H, DuttaRoy S, Rundqvist B, Börjesson M. The effect of physical activity or exercise on key biomarkers in atherosclerosis–a systematic review. Atherosclerosis. 2014;235(1):150-161. doi:10.1016/j.atherosclerosis.2014.04.026
- Sarzynski MA, Ruiz-Ramie JJ, Barber JL, et al. Effects of Increasing Exercise Intensity and Dose on Multiple Measures of HDL (High-Density Lipoprotein) Function. Arterioscler Thromb Vasc Biol. 2018;38(4):943-952. doi:10.1161/ATVBAHA.117.310307
- Kadoglou NP, Moustardas P, Kapelouzou A, et al. The anti-inflammatory effects of exercise training promote atherosclerotic plaque stabilization in apolipoprotein E knockout mice with diabetic atherosclerosis. Eur J Histochem. 2013;57(1):e3. Published 2013 Mar 25. doi:10.4081/ejh.2013.e3
- Brendel JM, Mayrhofer T, Karády J, et al. Risk in Women Emerges at Lower Coronary Plaque Burden Than in Men: PROMISE Trial. Circ Cardiovasc Imaging. 2026;19(3):e019011. doi:10.1161/CIRCIMAGING.125.019011
- Currie KD, Floras JS, La Gerche A, Goodman JM. Exercise Blood Pressure Guidelines: Time to Re-evaluate What is Normal and Exaggerated?. Sports Med. 2018;48(8):1763-1771. doi:10.1007/s40279-018-0900-x


