A Patient-Facing Q & A
Introduction
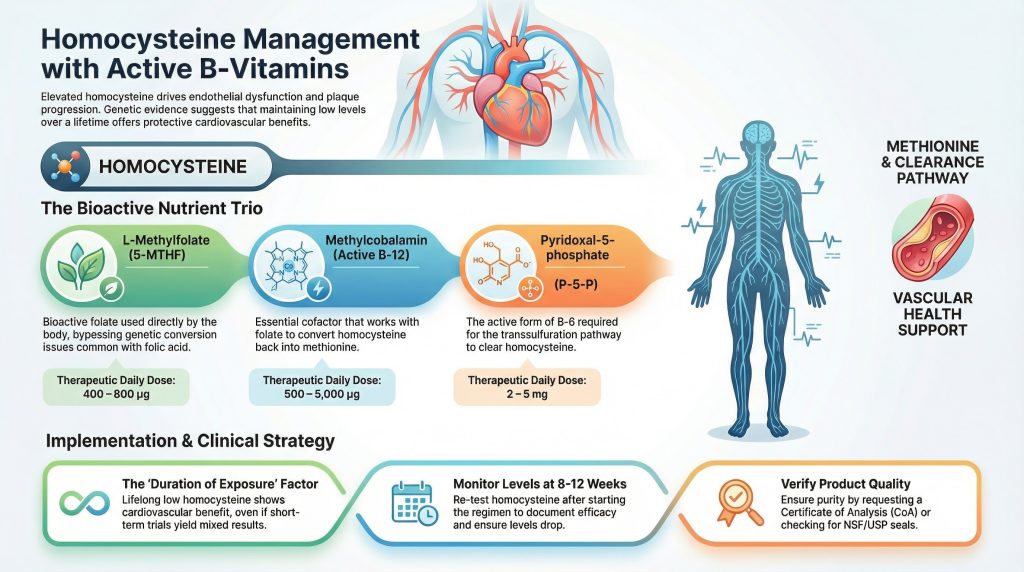
Cardiovascular prevention prioritizes proven interventions: cholesterol management, blood pressure control, smoking cessation, and lifestyle optimization. However, homocysteine is an additional modifiable risk factor—an amino acid linked to endothelial dysfunction and thrombosis. Supplements supplying the active cofactors for homocysteine metabolism—L-methylfolate (5-MTHF), methylcobalamin (active B-12), and pyridoxal-5-phosphate (P-5-P, active B-6)—reliably lower homocysteine levels. This Q&A explains the mechanism, the evidence regarding cardiovascular outcomes, product quality verification, and clinical integration.
Q & A
Q: What specific ingredients should I look for?
A: Look for supplements containing the active forms of the B-vitamins required for homocysteine metabolism:
- L-methylfolate (5-MTHF): The bioactive folate (often labeled as (6S)-5-MTHF or 5-methyltetrahydrofolate). Common doses: 400–800 µg.
- Methylcobalamin: The active form of B-12. Supplemental doses vary widely (e.g., 1,000–5,000 µg).
- Pyridoxal-5-phosphate (P-5-P): The active form of B-6. Modest doses (2–5 mg) are common.
Any brand listing these specific active ingredients at therapeutic doses adheres to the same clinical principles.
Q: Is “methylfolate” the same as L-methylfolate or 5-MTHF?
A: Yes. Labels may read L-methylfolate, 5-MTHF, 5-methyltetrahydrofolate, or (6S)-5-MTHF. These all refer to the bioactive form the body utilizes directly, unlike synthetic folic acid, which requires enzymatic conversion.
Q: How does this combination affect atherosclerosis or plaque?
A: Homocysteine is cleared via two primary biochemical pathways:
- Remethylation to methionine (requires 5-MTHF and methylcobalamin).
- Transsulfuration to cystathionine (requires P-5-P).
Elevated homocysteine drives endothelial dysfunction, oxidative stress, and prothrombotic changes—mechanisms accelerating plaque progression. Supplementing with active cofactors corrects metabolic bottlenecks, lowering homocysteine and mitigating this specific source of vascular injury.
Q: Will taking these vitamins prevent heart attacks and strokes?
A: Short answer: These supplements reliably lower homocysteine, but this does not always translate to reduced cardiovascular events in short-term clinical trials.
While large trials confirm predictable homocysteine reduction (often >20%), data on “hard outcomes” (heart attack, stroke) is mixed. Some trials found no reduction in composite endpoints despite lower homocysteine levels. However, specific subgroups—particularly those with low baseline folate or in stroke prevention contexts—have shown benefit. Therefore, use these supplements as part of a comprehensive plan, specifically when homocysteine is elevated, while maintaining proven therapies (statins, BP control, diet).
Q: Why do genetic studies show a risk, but some trials don’t show a cure? Is it a timing issue?
A: This is a leading scientific hypothesis known as the “duration of exposure.”
- Mendelian Randomization (Genetic Studies): Research looking at genetics shows that people born with efficient enzymes (who have naturally lower homocysteine their entire lives) have a significantly lower risk of heart disease compared to those with genetic variants that raise homocysteine. This suggests a causal link.
- The Time Gap: Clinical trials typically last only 3–5 years. Reversing decades of vascular damage caused by lifelong elevated homocysteine may require a longer duration than these trials allow.
- The Takeaway: While short-term vitamin therapy may not rapidly “scrub” existing plaque in advanced disease, the genetic data strongly suggests that keeping homocysteine low over the long term is protective for the vascular system.
Q: Are active forms (5-MTHF, methylcobalamin, P-5-P) superior to standard B-vitamins?
A: Generally, yes. Active forms are immediately bioavailable, bypassing potential absorption issues or genetic conversion defects (such as MTHFR variants). They offer a more predictable biochemical impact on homocysteine levels compared to synthetic folic acid or cyanocobalamin.
Q: Are the usual doses safe?
A: Typical therapeutic ranges are generally safe:
- 5-MTHF: 400–800 µg/day.
- Methylcobalamin: 500–5,000 µg/day (higher end is generally well-tolerated).
- P-5-P: 2–5 mg/day (safe range).
Note: Very high, long-term doses of B-6 (specifically pyridoxine) can cause neuropathy. Remain within recommended limits unless medically supervised.
Q: Who should consult a clinician first?
A: Consult a provider if you are pregnant/breastfeeding, have chronic kidney or liver disease, have a history of cancer, or take medications interacting with folate (e.g., methotrexate, anticonvulsants). Unexplained neuropathy or anemia also requires medical evaluation before supplementation.
Q: What monitoring is recommended?
A:
- Baseline: Plasma homocysteine, serum B-12 (± methylmalonic acid), serum/RBC folate, lipid panel, and blood pressure.
- Follow-up (8–12 weeks): Recheck homocysteine to document efficacy.
- Non-responders: If homocysteine remains elevated, review adherence, absorption issues (e.g., celiac/GI disease), renal function, and drug interactions.
Q: How can I verify product quality?
A:
- Certifications: Look for USP, NSF, ConsumerLab, or Informed-Choice seals.
- Independent Testing: Check reports from ConsumerLab or Labdoor.
- Certificate of Analysis (CoA): Request a CoA from the manufacturer to verify potency and purity (heavy metals/microbes).
Generic CoA Request Template:
Subject: Request for Certificate of Analysis (CoA) — [Product Name & Lot #]
To [Manufacturer Name] Quality Assurance,
I recently purchased [Product Name] (Lot #: ____). For clinical review, please provide the Certificate of Analysis (CoA) for this specific lot. I require verification of assay results for L-methylfolate (5-MTHF), methylcobalamin, and P-5-P, as well as screening results for heavy metals and contaminants.
Thank you,
[Your Name]
Clinician’s Implementation Strategy
- Assess: Measure baseline risks and labs.
- Prioritize: Optimize proven therapies (statins, BP control, Mediterranean diet, exercise).
- Treat: If homocysteine is elevated, initiate active-form regimen (5-MTHF + Methyl B-12 + P-5-P).
- Monitor: Recheck homocysteine at 8–12 weeks.
- Contextualize: Use as an adjunctive biochemical tool, not a replacement for standard preventive care.
Clinical Sidebar: Checklist
- [ ] Baseline Labs: Hcy, B-12, Folate, Lipids, BP.
- [ ] Intervention: Active-form B-complex if Hcy elevated.
- [ ] Follow-up: Re-test Hcy at 8–12 weeks.
- [ ] Audit: If no response, check absorption/compliance.
Bottom Line
Supplements providing L-methylfolate, methylcobalamin, and P-5-P offer a rational, evidence-based method to lower homocysteine. While short-term trials have been mixed, genetic (Mendelian) studies suggest that lifelong lower homocysteine correlates with reduced cardiovascular risk. They are best utilized within a broad atherosclerosis prevention strategy that prioritizes proven lipid and blood pressure management. Always verify product quality and monitor blood markers to ensure efficacy.
References
- Lonn E, Yusuf S, Arnold MJ, et al. Homocysteine lowering with folic acid and B vitamins in vascular disease. N Engl J Med. 2006;354(15):1567-1577. doi:10.1056/NEJMoa060900
- Spence JD, Howard VJ, Chambless LE, et al. Vitamin Intervention for Stroke Prevention (VISP) trial: rationale and design. Neuroepidemiology. 2001;20(1):16-25. doi:10.1159/000054753
- Bønaa KH, Njølstad I, Ueland PM, et al. Homocysteine lowering and cardiovascular events after acute myocardial infarction. N Engl J Med. 2006;354(15):1578-1588. doi:10.1056/NEJMoa055227
- Wald DS, Law M, Morris JK. Homocysteine and cardiovascular disease: evidence on causality from a meta-analysis. BMJ. 2002;325(7374):1202. doi:10.1136/bmj.325.7374.1202
- Martí-Carvajal AJ, Solà I, Lathyris D, Dayer M. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst Rev. 2017;8(8):CD006612. Published 2017 Aug 17. doi:10.1002/14651858.CD006612.pub5


