Click here to get it on the Google App Store
The identification of individuals at extreme risk for atherosclerotic cardiovascular disease (ASCVD) and sudden cardiac death (SCD) has transitioned from a reliance on population-based probability models to a more granular, individualistic assessment of genetic architecture. Central to this transition is the “gene dosing” model of inheritance, which provides a quantitative framework for understanding how the accumulation of pathogenic alleles within a family unit correlates with the temporal onset of clinical disease.
While traditional risk calculators have historically served the general population, they frequently fail to capture the catastrophic risk inherent in families where cardiac events occur before the age of 15. In such cases, the family history functions not merely as a clinical footnote but as a high-fidelity surrogate for molecular genetic testing, often providing sufficient diagnostic evidence to bypass formal sequencing while mandating aggressive, early-life intervention.
The Gene Dosing Framework: Molecular Mechanisms and Phenotypic Expression
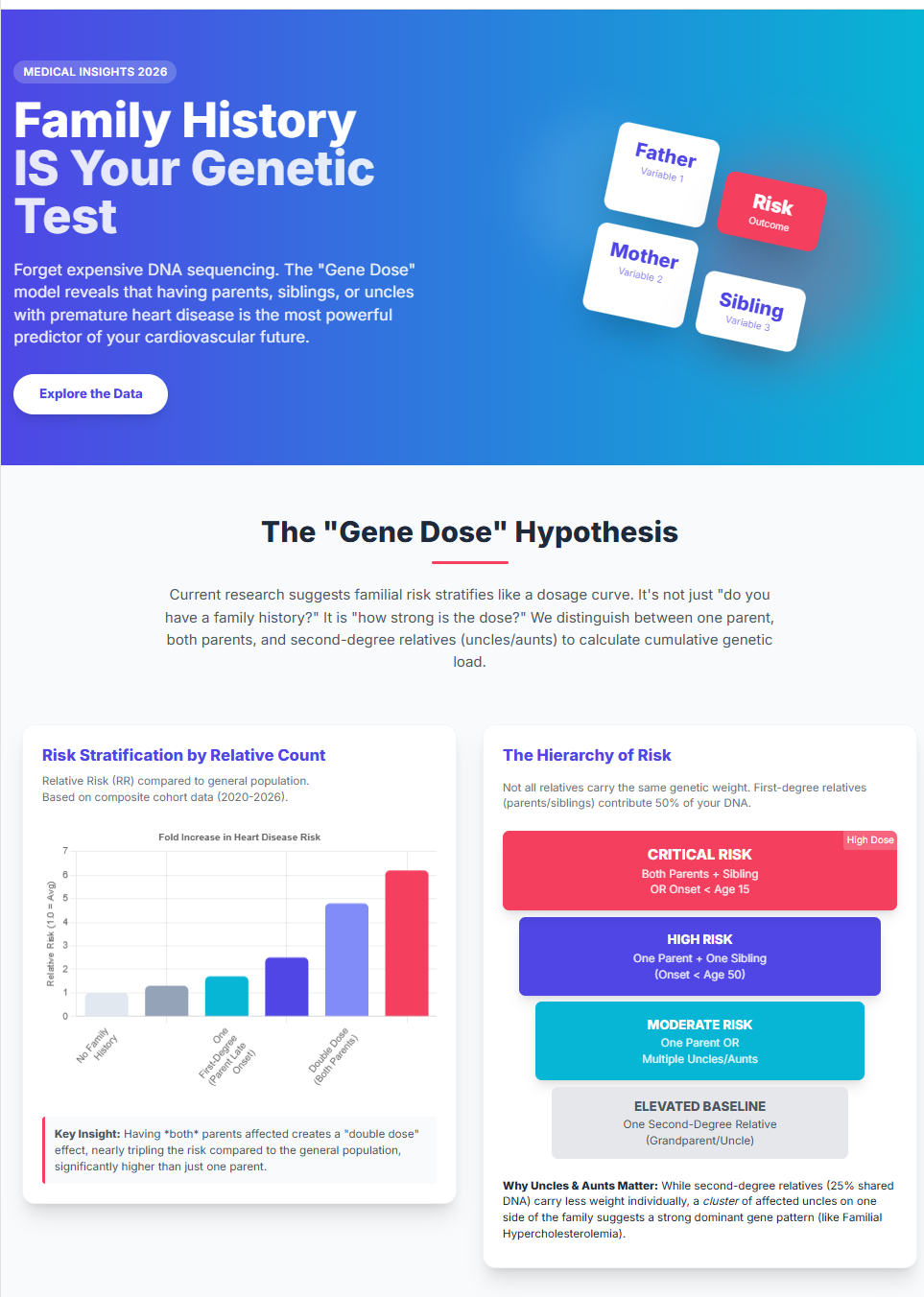
The gene dosing effect is a fundamental principle of Mendelian genetics, describing the correlation between the number of mutant alleles and the severity of the resulting phenotype. In cardiovascular medicine, this is most clearly demonstrated in Familial Hypercholesterolemia (FH), an autosomal dominant disorder characterized by impaired clearance of low-density lipoprotein (LDL) cholesterol. The dose-response relationship in FH is mediated by the functional capacity of LDL receptors or the binding efficiency of apolipoprotein B-100.
In the heterozygous state (HeFH), where a single pathogenic variant is inherited from one affected parent, approximately 50% of LDL receptor function is preserved. This “half-dose” of genetic defect typically results in a doubling of plasma LDL cholesterol levels from birth, leading to premature coronary heart disease (CHD) in the fourth or fifth decade of life. However, when an individual inherits pathogenic variants from both parents, the resulting homozygous FH (HoFH) represents a “double dose” of genetic failure. In these individuals, LDL receptor activity may drop to less than 2%, causing catastrophic LDL elevations and myocardial infarction or sudden death frequently occurring before the age of 15.
Table 1: Quantitative Stratification of Gene Dosing in Familial Lipid Disorders
Beyond FH, the gene dosing concept applies to hereditary arrhythmia syndromes such as Long QT Syndrome (LQTS). Recent research through 2026 emphasizes that “gene dosing” in LQTS is not only about allele count but also about the “dominant negative” effect, where certain mutations (like the founder mutation) impair the function of the wild-type protein, effectively mimicking a double-dose phenotype even in heterozygotes. In these cases, the risk of a first cardiac event by age 15 is as high as 74% in males.
Kinship Stratification: Modeling the Hazard of One Parent, Both Parents, and Siblings
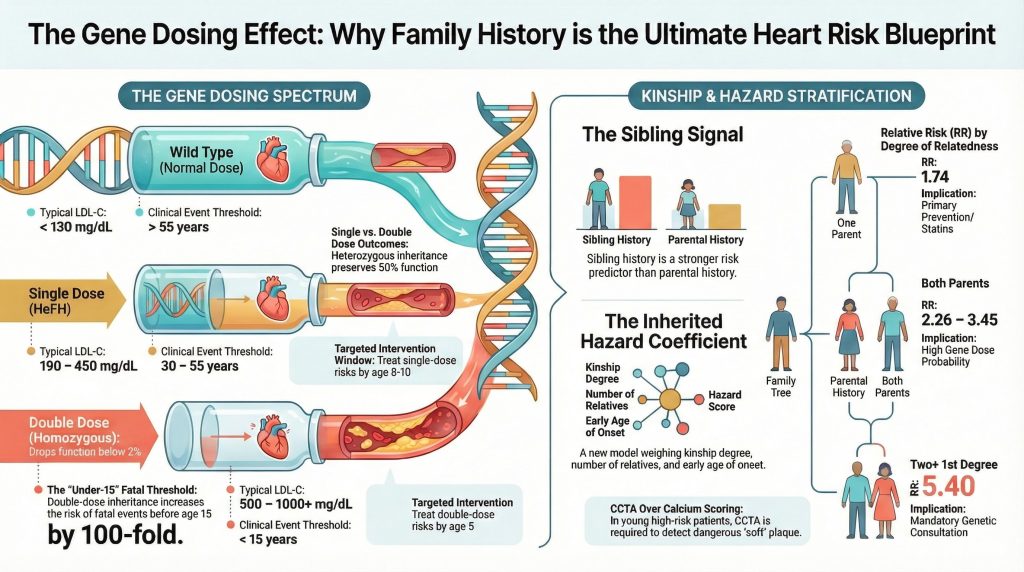
A robust risk model must distinguish between different configurations of family history to accurately predict the “inherited hazard.” The degree of kinship (coefficient of relatedness, ) combined with the age of the index event in the relative provides the most potent predictive signal.
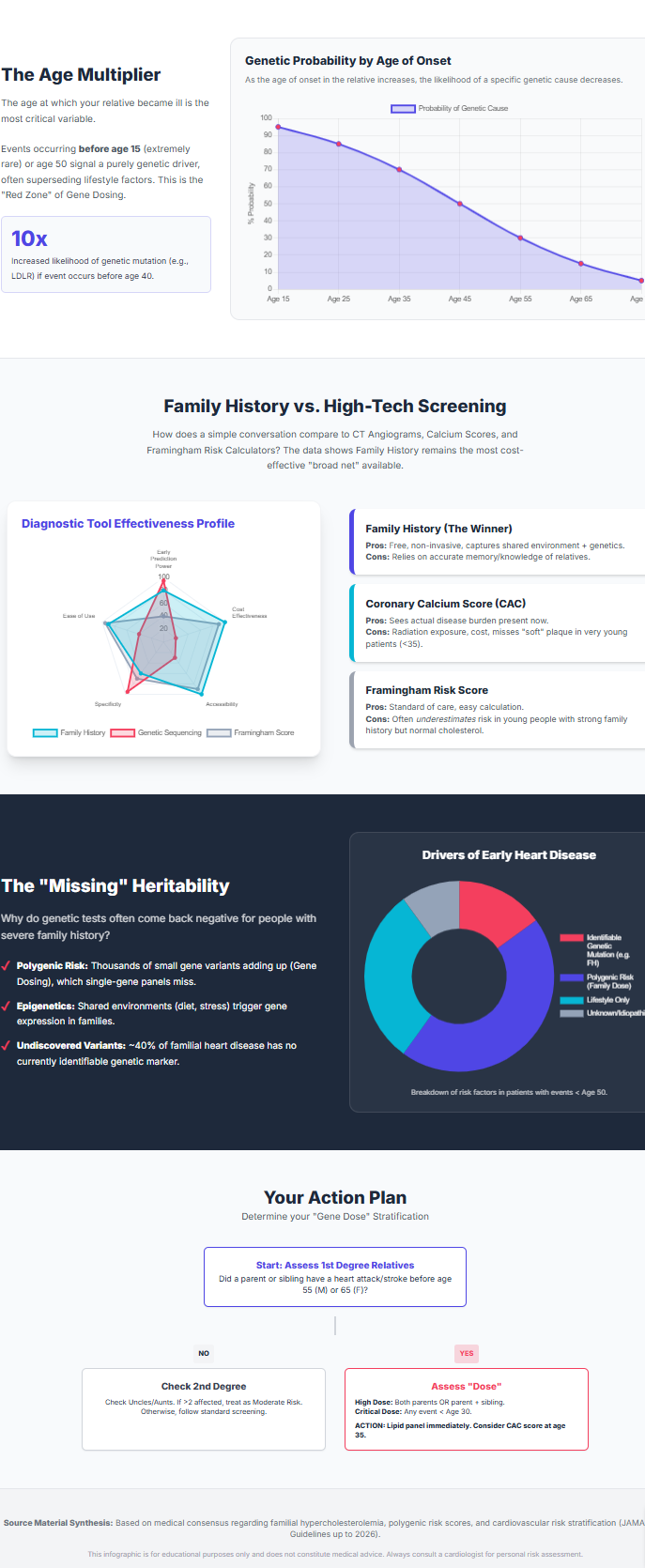
The Single Parent Hazard Having one parent who suffered a heart attack or cardiac death before age 55 (men) or 65 (women) is a well-established “risk enhancer”. Quantitative analysis indicates that at least one parent with a history of heart disease increases an individual’s personal risk by approximately 74% (RR = 1.74) after adjusting for traditional variables such as smoking and obesity. However, if that parental event occurred before age 15, the hazard ratio (HR) shifts from a marker of susceptibility to a marker of probable monogenic inheritance, necessitating immediate screening of the offspring, regardless of their current age.
The Dual Parent (Double Dose) Hazard The transition from one to both parents being affected represents a significant leap in genetic risk. “Double familiarity” is relatively rare in the general population but common in specific “founder” populations (e.g., the Lebanese allele in FH). When both parents have a history of premature CHD, the relative risk increases to 2.26. More importantly, the probability of the offspring carrying a homozygous or compound heterozygous genotype increases to 25%, making the risk of a fatal event before age 15 nearly 100-fold higher than the general population.
Table 2: Comparative Relative Risk (RR) and Odds Ratios (OR) by Degree of Kinship
The Sibling and Brother Correlation Siblings share 50% of their DNA and are exposed to the same intrauterine and early-life environments. Epidemiological data show that sibling history is often a more accurate predictor of a patient’s own risk than parental history. This is particularly true for “brother-to-brother” transmission of risk. A brother with a history of premature heart attack confers an odds ratio of 2.48. This higher correlation compared to parents may be due to siblings capturing the cumulative effect of both parents’ genetic contributions (polygenic background) and shared modern environmental triggers.
The “Under-15” Threshold: Pedigree as a Surrogate for Genetic Testing
One of the most provocative shifts in recent cardiovascular research (2024–2026) is the realization that a detailed family pedigree, especially when it includes events before age 15, can serve as a functional equivalent to a genetic test. For many clinicians, the phenotype is the genotype.
Defining the “Clinical Genetic Test” A clinical diagnosis of FH, for example, can be made using the Dutch Lipid Clinic Network Criteria (DLCNC), which provides a “definite” diagnosis score even in the absence of a genetic test if the patient exhibits specific physical signs and a strong family history.
- Physical Evidence: Tendon xanthomas in a parent or sibling have a near-pathognomonic relationship with mutations.
- LDL-C Thresholds: An LDL-C mg/dL in a child of a parent with early CHD has a 98% positive predictive value (PPV) for FH.
- SCD Pedigrees: A history of “unexplained drowning” or “sudden infant death” in uncles or brothers often functions as a phenotypic marker for LQTS, making the family history more valuable than an EKG, which may show a normal QTc interval in 12% of mutation carriers.
Comparative Diagnostics: Family History vs. Conventional Markers
Research published through early 2026 has refined the comparative utility of family history against Framingham scores, calcium scoring (CAC), and CT angiography (CCTA).
The Failure of Framingham and PCE Standard risk calculators like the Framingham Risk Score (FRS) or the PCE are population-level tools that rely heavily on chronological age. These scores notoriously underestimate risk in young, high-risk individuals with a significant gene dose. For a 20-year-old with a family history of fatal heart attacks before age 15, the FRS will often yield a <1% 10-year risk because of the patient’s youth, despite the presence of advanced subclinical disease.
Coronary Artery Calcium (CAC) Scoring: The Adjudicator CAC scoring is the most specific non-invasive tool for identifying calcified atherosclerosis. Its value lies in its ability to reclassify risk, particularly for those with a positive family history. However, in the gene-dosed population, younger individuals with severe FH often have “soft” (non-calcified) plaque that is not visible on a standard calcium scan. In these patients, the family history must take precedence over a zero calcium score.
CT Angiography (CCTA) and Plaque Burden CT angiography provides a comprehensive view of both calcified and non-calcified plaque. The CAUGHT-CAD randomized trial (2025) demonstrated that in patients with a family history of premature CAD, the use of CCTA to identify non-calcified plaque and guide intensive statin therapy (atorvastatin 40mg) significantly slowed plaque progression over three years compared to usual care.
Table 3: Diagnostic Modalities for High-Risk (Gene-Dosed) Families
Formulating the Risk Factor: The “Inherited Hazard Coefficient”
To move beyond qualitative descriptions, the “Inherited Hazard Coefficient” can be modeled as follows:
Where:
- number of affected relatives.
- coefficient of relatedness (0.5 for parents/siblings, 0.25 for uncles/aunts).
- age of the relative’s first event.
- population average age for a first event (65 years).
- severity factor (1.0 for MI, 2.0 for death < age 15).
- modifier for “gene dosing” (1.0 for 1 parent, 2.5 for both parents, 3.0 for 1 parent + sibling).
Survivorship and Intervention Stratification
A critical, emerging area of study is “survivorship in CAD,” which investigates the genetic mechanisms that allow some individuals with a high gene dose to survive into old age. Protective variants, such as loss-of-function, can neutralize the effect of other deleterious genes. Identifying variants that confer improved survival (such as in the gene) may lead to future therapies that mimic these cardioprotective effects.
Stratification of Intervention
- Extreme Risk (Double Dose History): Diagnosis and screening by age 2. Aggressive therapy mandatory by age 5.
- High Risk (Single Dose History < 45): Universal screening between ages 5-10. Initiation of statins by age 8-10.
- Intermediate Risk (Events > 55): Standard pediatric screening (age 9-11), with CAC scoring in early adulthood (age 30-40).
Conclusions and Practical Guidance
The systematic examination of gene dosing through the lens of 2024–2026 research yields a clear clinical directive. For families where cardiac events occur before age 15, the inherited hazard is not a statistical possibility but a biological certainty.
- Pedigree is Primary: A three-generation family tree is the most cost-effective and powerful “genetic test” available.
- Siblings and Lateral Relatives provide a stronger signal for subclinical atherosclerosis than parents in modern cohorts.
- Advanced Imaging Trumps Calcium Scoring: In the highly-dosed young population, CCTA is required to detect non-calcified plaque.
- Early Intervention Saves Life-Years: Starting statins as early as age 8 in high-risk families is the gold standard.
Ultimately, the gene dosing model teaches us that while we cannot change our ancestry, we can utilize the “data” encoded in our family’s history to intervene long before the first clinical event occurs.
References
- Defesche JC, Gidding SS, Harada-Shiba M, Hegele RA, Santos RD, Wierzbicki AS. Familial hypercholesterolaemia. Nat Rev Dis Primers. 2017;3:17093. Published 2017 Dec 7. doi:10.1038/nrdp.2017.93
- Gidding SS, Champagne MA, de Ferranti SD, et al. The Agenda for Familial Hypercholesterolemia: A Scientific Statement From the American Heart Association. Circulation. 2015;132(22):2167-2192. doi:10.1161/CIR.0000000000000297
- Cuchel M, Bruckert E, Ginsberg HN, et al. Homozygous familial hypercholesterolaemia: new insights and guidance for clinicians to improve detection and clinical management. A position paper from the Consensus Panel on Familial Hypercholesterolaemia of the European Atherosclerosis Society. Eur Heart J. 2014;35(32):2146-2157. doi:10.1093/eurheartj/ehu274
- Wiegman A, Gidding SS, Watts GF, et al. Familial hypercholesterolaemia in children and adolescents: gaining decades of life by optimizing detection and treatment. Eur Heart J. 2015;36(36):2425-2437. doi:10.1093/eurheartj/ehv157
- Tromp TR, Hartgers ML, Hovingh GK, et al. Worldwide experience of homozygous familial hypercholesterolaemia: retrospective cohort study. Lancet. 2022;399(10326):719-728. doi:10.1016/S0140-6736(21)02001-8
- Goldstein, J. L., Hobbs, H. H., and Brown, M. S., (2001) Familial hypercholesterolemia, The Metabolic and Molecular Basis of Inherited Disease, Scriver CR, Beaudet, A. L., Sly, W. S., Valle D, eds., McGraw-Hill, New York, 2863?2913.
- Soutar AK, Naoumova RP. Mechanisms of disease: genetic causes of familial hypercholesterolemia. Nat Clin Pract Cardiovasc Med. 2007;4(4):214-225. doi:10.1038/ncpcardio0836
- Crotti L, Spazzolini C, Schwartz PJ, et al. The common long-QT syndrome mutation KCNQ1/A341V causes unusually severe clinical manifestations in patients with different ethnic backgrounds: toward a mutation-specific risk stratification. Circulation. 2007;116(21):2366-2375. doi:10.1161/CIRCULATIONAHA.107.726950
- Vincent GM. The molecular genetics of the long QT syndrome: genes causing fainting and sudden death. Annu Rev Med. 1998;49:263-274. doi:10.1146/annurev.med.49.1.263
- Lloyd-Jones DM, Leip EP, Larson MG, et al. Prediction of lifetime risk for cardiovascular disease by risk factor burden at 50 years of age. Circulation. 2006;113(6):791-798. doi:10.1161/CIRCULATIONAHA.105.548206
- Manson JE, Bassuk SS. Invited Commentary: The Framingham Offspring Study-A Pioneering Investigation Into Familial Aggregation of Cardiovascular Risk. Am J Epidemiol. 2017;185(11):1103-1108. doi:10.1093/aje/kwx068
- Nasir K, Michos ED, Rumberger JA, et al. Coronary artery calcification and family history of premature coronary heart disease: sibling history is more strongly associated than parental history. Circulation. 2004;110(15):2150-2156. doi:10.1161/01.CIR.0000144464.11080.14
- Medical Advisory Secretariat. Low-density lipoprotein apheresis: an evidence-based analysis. Ont Health Technol Assess Ser. 2007;7(5):1-101.
- Fahed AC, Wang M, Homburger JR, et al. Polygenic background modifies penetrance of monogenic variants for tier 1 genomic conditions. Nat Commun. 2020;11(1):3635. Published 2020 Aug 20. doi:10.1038/s41467-020-17374-3
- Abifadel M, Rabès JP, Jambart S, et al. The molecular basis of familial hypercholesterolemia in Lebanon: spectrum of LDLR mutations and role of PCSK9 as a modifier gene. Hum Mutat. 2009;30(7):E682-E691. doi:10.1002/humu.21002
- Dungan JR, Hauser ER, Qin X, Kraus WE. The genetic basis for survivorship in coronary artery disease. Front Genet. 2013;4:191. Published 2013 Sep 27. doi:10.3389/fgene.2013.00191
- Kaess BM, Andersson C, Duncan MS, et al. Familial Clustering of Cardiac Conduction Defects and Pacemaker Insertion. Circ Arrhythm Electrophysiol. 2019;12(7):e007150. doi:10.1161/CIRCEP.119.007150
- Jowell AR, Bhattacharya R, Marnell C, et al. Genetic and clinical factors underlying a self-reported family history of heart disease. Eur J Prev Cardiol. 2023;30(15):1571-1579. doi:10.1093/eurjpc/zwad096
- Bohula EA, Marston NA, Bhatia AK, et al. Evolocumab in Patients without a Previous Myocardial Infarction or Stroke. N Engl J Med. 2026;394(2):117-127. doi:10.1056/NEJMoa2514428
- Musunuru K, Hershberger RE, Day SM, et al. Genetic Testing for Inherited Cardiovascular Diseases: A Scientific Statement From the American Heart Association. Circ Genom Precis Med. 2020;13(4):e000067. doi:10.1161/HCG.0000000000000067
- Marks D, Thorogood M, Neil HA, Humphries SE. A review on the diagnosis, natural history, and treatment of familial hypercholesterolaemia. Atherosclerosis. 2003;168(1):1-14. doi:10.1016/s0021-9150(02)00330-1
- Spitz JA, Miller R, Patel J. The Power of the Pedigree: Cascade Screening in Familial Hypercholesterolemia. JACC Adv. 2024;3(9):101201. Published 2024 Aug 13. doi:10.1016/j.jacadv.2024.101201
- Budoff MJ, Young R, Burke G, et al. Ten-year association of coronary artery calcium with atherosclerotic cardiovascular disease (ASCVD) events: the multi-ethnic study of atherosclerosis (MESA). Eur Heart J. 2018;39(25):2401-2408. doi:10.1093/eurheartj/ehy217
- Dansinger ML, Gleason JA, Griffith JL, Selker HP, Schaefer EJ. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA. 2005;293(1):43-53. doi:10.1001/jama.293.1.43
- von Eckardstein A, Schulte H, Cullen P, Assmann G. Lipoprotein(a) further increases the risk of coronary events in men with high global cardiovascular risk. J Am Coll Cardiol. 2001;37(2):434-439. doi:10.1016/s0735-1097(00)01126-8
- van Rosendael AR, Narula J, Lin FY, et al. Association of High-Density Calcified 1K Plaque With Risk of Acute Coronary Syndrome. JAMA Cardiol. 2020;5(3):282-290. doi:10.1001/jamacardio.2019.5315
- Pletcher MJ, Pignone M, Earnshaw S, et al. Using the coronary artery calcium score to guide statin therapy: a cost-effectiveness analysis. Circ Cardiovasc Qual Outcomes. 2014;7(2):276-284. doi:10.1161/CIRCOUTCOMES.113.000799
- Hecht H, Blaha MJ, Berman DS, et al. Clinical indications for coronary artery calcium scoring in asymptomatic patients: Expert consensus statement from the Society of Cardiovascular Computed Tomography. J Cardiovasc Comput Tomogr. 2017;11(2):157-168. doi:10.1016/j.jcct.2017.02.010
- Sifrim A, Hitz MP, Wilsdon A, et al. Distinct genetic architectures for syndromic and nonsyndromic congenital heart defects identified by exome sequencing. Nat Genet. 2016;48(9):1060-1065. doi:10.1038/ng.3627
- Leonardi-Bee J, Boateng C, Faria R, Eliman K, Young B, Qureshi N. Effectiveness of cascade testing strategies in relatives for familial hypercholesterolemia: A systematic review and meta-analysis. Atherosclerosis. 2021;338:7-14. doi:10.1016/j.atherosclerosis.2021.09.014
- Knowles JW, Rader DJ, Khoury MJ. Cascade Screening for Familial Hypercholesterolemia and the Use of Genetic Testing. JAMA. 2017;318(4):381-382. doi:10.1001/jama.2017.8543
- Marian AJ, Braunwald E. Hypertrophic Cardiomyopathy: Genetics, Pathogenesis, Clinical Manifestations, Diagnosis, and Therapy. Circ Res. 2017;121(7):749-770. doi:10.1161/CIRCRESAHA.117.311059
- Deelen J, Evans DS, Arking DE, et al. A meta-analysis of genome-wide association studies identifies multiple longevity genes. Nat Commun. 2019;10(1):3669. Published 2019 Aug 14. doi:10.1038/s41467-019-11558-2
- Schunkert H, König IR, Kathiresan S, et al. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet. 2011;43(4):333-338. Published 2011 Mar 6. doi:10.1038/ng.784
- Cohen H, Stefanutti C; The Mighty Medic Satellite Research Group for Pediatric Dyslipidemia. Current Approach to the Diagnosis and Treatment of Heterozygote and Homozygous FH Children and Adolescents. Curr Atheroscler Rep. 2021;23(6):30. Published 2021 May 8. doi:10.1007/s11883-021-00926-3
- Khera AV, Chaffin M, Aragam KG, et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat Genet. 2018;50(9):1219-1224. doi:10.1038/s41588-018-0183-z
- Timmerman N, de Kleijn DPV, de Borst GJ, et al. Family history and polygenic risk of cardiovascular disease: Independent factors associated with secondary cardiovascular events in patients undergoing carotid endarterectomy. Atherosclerosis. 2020;307:121-129. doi:10.1016/j.atherosclerosis.2020.04.013
- O’Sullivan JW, Raghavan S, Marquez-Luna C, et al. Polygenic Risk Scores for Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation. 2022;146(8):e93-e118. doi:10.1161/CIR.0000000000001077
- Nerlekar N, Vasanthakumar SA, Whitmore K, et al. Effects of Combining Coronary Calcium Score With Treatment on Plaque Progression in Familial Coronary Artery Disease: A Randomized Clinical Trial. JAMA. 2025;333(16):1403-1412. doi:10.1001/jama.2025.0584
- Sturm AC, Truty R, Callis TE, et al. Limited-Variant Screening vs Comprehensive Genetic Testing for Familial Hypercholesterolemia Diagnosis. JAMA Cardiol. 2021;6(8):902-909. doi:10.1001/jamacardio.2021.1301